(First upload on April 11 2004. Last on September 4 2018) [ 日本語 | English ]
Mount Usu / Sarobetsu post-mined peatland
From left: Crater basin in 1986 and 2006. Cottongrass / Daylily
HOME > Plant list (植物リスト) > Pinaceae (マツ科) > Larix kaempferi
[ Larix | L. kaempferi | L. decidua | L. gmelinii | L. olgensis ]
Larix Mill. (カラマツ)Nihon-Karamatsu (ニホンカラマツ, 日本落葉松), Japanese larchLifeform: tall deciduous tree < 50 m high Distribution: native to central and northern Japan (but non-native to Hokkaido) transplanted to Hokkaido after Meiji 30s' |
Habitat: sunny and dry sites (by high morphological plasticity (Akasaka & Tsuyuzaki 2005) Seed dispersal: wind Synonyms: Larix leptolepis (Siebold et Zucc.) Gordon f. pendula (Sugim.) Yonek. (シダレカラマツ)f. rubescens (Inokuma) T. Shimizu (アカミカラマツ) |
|
 1
1
 2
2
 3
3
 4
4
 5
5
[1/2, 4/5] taken on the middle slope of Mount Koma. [1] in October 2001, by M. Akasaka. [2] in the summer of 2001 by Jon. [3] a bark (樹皮) in the Arboretum of Faculty of Agriculture, Hokkaido University, close to Poplar Street, on April 19 2014. [4] a spur shoot on May 1 2017. [5] on July 9 2009. The cone production was high in this year.
Key words: biological invasion (Kondo & Tsuyuzaki 1999), exotic or non-native plant (Titus & Tsuyuzaki 2003), facilitation (Uesaka & Tsuyuzaki 2004)
 1
1
 2
2
 3
3
[1-3] in front of Date City Office, close to Mount Usu, on July 25 2018.
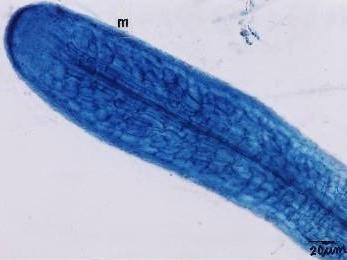 |
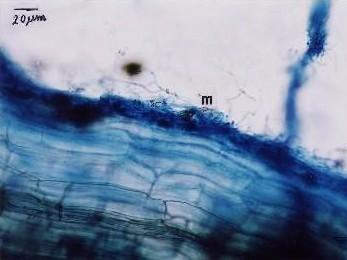 |
Fig. Ectomycorrhizae from the roots of L. kaempferi. M: mantle. The sample was collected from the southwest slope of Mount Koma Tsuyuzaki et al. 2005. Mycorrhizal fungi grew in the roots even at the seedling stage (Akasaka & Tsuyuzaki 2007). |
 A canopy at Spaskaya Pad, Sakha Republic, on July 16 1995. |
 A woodland near Kolyma River on July 14 1995. Russian scientists often adopt L. cajanderi. |
Syn. L. dahurica (Loudon) Turcz. ex Trautv. var. japonica Maxim. ex Regel, L. kurilensis Mayr Habitat/distribution: dominant species in Siberian taiga Varietiesvar. gmeliniivar. japonica (Maxim. ex Regel) Pilg. var. olgensis (A. Henry) Ostenf. et Syrach → L. olgensis A. Henry] var. principis-rupprechtii (Mayr) Pilg.
|
|
Chousenkaramatsu (チョウセンカラマツ, 朝鮮落葉松), Olga Bay larch
黄花落叶松 in Chinese Distribution: northern Korea and its neighbouring areas |
Mount Changbai: 500-1900 m elevation Habitat: the margins of swamps and lakesBark (樹皮) |
Synonyms:
Larix gmelinii Rupr. ex Kuzen. var. olgensis (A. Henry) Ostenf. et Syrach f. viridis (E. H. Wilson) Nakai (アオミノチョウセンカラマツ) |
 1
1
 2
2
 3
3
[1/2] at the arboretum of Hokkaido Research Center, Forestry and Forest Products Research Institute, in Hitsujigaoka, Sapporo, on July 9 2013. [3] in the Dell Forest, Changbai Mountain, Jilin Province, China, on June 12 2017.
Structure of a thinned Larix olgensis forest in Sandaohu peatland, Jiling Province, ChinaShiro Tsuyuzaki Graduate School of Science and Technology Niigata University Niigata 950-21 Japan Vegetation in China has been influenced by large-scaled cultivation including logging for more than 3000 years (Hou, 1983). Despite this intensive land use, the structure and management of forests in Chinese peatlands have rarely been reported (Chai, 1980). Forests dominated by Larix olgensis Henry are widespread in the peatlands of northeastern China, including Sandaohu, Jiling Province, China (42°80'N, 126°90'E). In this location, peatland forests have been subjected to logging and are threatened by a drainage project under construction. I report the structure of a thinned L. olgensis forest, to document changes after logging and prior to drainage. The survey was conducted October 1989 in a peatland in Sandaohu. The climate is moist, microthermal to mesothermal with a mean annual temperature of -4 to 4°C (Chai, 1980). Water level was -20 to 0 cm in the center of the peatland during the 10-day survey period (T. Nagasawa, personal communication). A 15 m × 15 m plot was set up in an area where L. olgensis was dominant. All stems in the plot were identified to species, mapped and measured for height, diameter at breast height (DBH), canopy width, and age using annual-ring counts. Cores were removed at a height of 20 cm and the width of each annual ring was measured under a binocular stereomicroscope to the nearest 0.01 mm. Trees whose crowns entered the plot but stems were outside were also recorded for location, height and DBH. The proportions of light gaps and crown overlaps were determined based on a map of crowns using a digitizer (Graphtec, KD-6400). The locations of cut stumps were mapped, and the height and basal diameter of each stump were measured. On the basis of stem locations, spatial distribution patterns were estimated by the Iδ index, a method which distinguishes three spatial distribution patterns, - random, uniform, and contiguous (Morisita, 1959). This index also determines the scale of aggregated patches in the case of a contiguous distribution. Approximately six ha around the plot were surveyed to determine seedling establishment and forest floor plant cover. In this area, I randomly set up 24 10 m × 10 m quadrat, and recorded maximum height of trees, cover of shrub layers (2-8 m in height), the total cover of herbs and mosses, and evidence of thinning. Peat cores were extracted, using a 1-m long peat sampler, to evaluate peat thickness and to determine the occurrence of flooding which is indicated by the traces of gravel and/or sand layers in peat. |
Maximum tree height was less than 20 m in the 6 ha surveyed peatland. Peat thickness was less than 20 cm deep in the forest. Gravel/sand layers were observed in most peat cores but were illegible, suggesting that the flooding occurred in the past but that the periodicity was unclear (M. Kamiya and T. Nagasawa, personal communication). The dominant tree species was L. olgensis. Betula platyphylla was also present. Shrub layers consisting of L. olgensis were sparse. The forest floor was mostly covered by grasses such as Phragmites communis, sedges, and mosses (Sphagnum spp.). Herbaceous plant cover was less than 60%, and moss cover was less than 10%. The total cover of mosses and herbs was usually about 40%, and never more than 70%. Therefore, vegetation structure was roughly homogeneous. Thinning intensity was similar throughout the forest. No tree seedlings were recorded in the plot and few in the adjacent 6 ha, although a large quantity of cones were produced on the branches of L. olgensis. Forty-six trees were recorded in the plot; 41 L. olgensis (89.1% of the total), four B. platyphylla (8.7%) and one Populus koreana (2.2%). Ten stems of L. olgensis were recorded near the plot. L. olgensis showed a wide range in height (10.6 ± 3.7 m, range; 1.8-14.9) and DBH (12.2 ± 5.6 cm, 0.9-22.3). The age of the 41 L. olgensis stems ranged from 20 to 39 years (30.9 ± 0.7 years). The age distribution of cut stumps was similar to that of standing stems, i.e., from 13 to 21 years. Twenty-six of the L. olgensis stems were aged at 30-34 years, and an additional nine stems were 26-29 years old. The height and DBH of L. olgensis were strongly correlated (r = +0.909, p <0.001), as were the relationships between age and height (r = +0.747, p <0.001) and age and DBH (r = +0.692, p <0.001). The age of short trees (<10 m) were from 20 to 28 years. Maximum crown width was 5 m. Crowns occupied 67.8% of the space, and various-sized light gaps occupied 32.2%. Twenty-three cut stumps were found in the plot. It is assumed that few stumps disappeared due to rot. Therefore, 1/3 of the original 64 tree stems were thinned. Morisita's Iδ index showed that the distribution patterns of trees changed from small contiguous patches, ranging in size from 0.04 to 3 m², before the thinning to a random pattern after the thinning. The small patches suggest that most crowns greatly overlapped before thinning. At the time of survey, only 12% of the crowns were overlapped. The change in distribution pattern and decrease of crown overlap in the present suggests that the thinning was mostly undertaken in the high density sites and the creation of crown gaps was due to thinning. On the basis of the personal communication of the foresters, it was confirmed that thinning was carried out 10-18 years ago. Mean annual-ring width gradually increased from 0.9 to 2.6 mm before the thinning, i.e, more than 19 years ago, and ranged from 2.8 to 3.3 mm during the logging period, i.e., 10-18 years ago. The largest mean annual ring width was 3.3 mm and was recorded eleven years ago. However, ring widths decreased soon after logging. For the last 10 years, annual ring widths were less than 2.0 mm. The post logging decrease in growth indicated that the logging had little or no influence on stem width growth. |
Even though thinning created crown gaps and decreased contiguous patches of overlapping crowns, the influence on stem growth was minor. Spatial competition for light between trees is a major limiting factor on growth (Tilman, 1988; Urban and Shugart, 1992). Because logging decreases crown overlaps, decreases light competition and subsequently promotes stem growth, logging generally increases annual-ring growth (Oliver and Larson, 1990). However, L. olgensis seems not to have responded to the increased light environment created by the thinning. In wetlands, excessive water supply leads to soil deoxidization and limits forest development (Heinselman, 1975). Thick accumulations of peat also inhibit plant growth (Bertness, 1988; Yabe, 1989). Peat depth influenced the presence of L. olgensis in the 6 ha surveyed. L. olgensis and B. platyphylla dominated sites where peat is 3-20 cm deep, but L. olgensis was absent from sites where peat was > 40 cm thick. This is similar to black spruce-feathermoss forests surrounding bogs in Minnesota (Heinselman, 1975). Instead of the light resource, therefore, soil conditions such as deoxidization and anaerobic soil, chemical composition, and water levels are likely to have an important role on the regeneration of L. olgensis forest. No tree seedlings were observed in surveyed areas despite high cone production, suggesting that the present environment is not suitable for seed germination and/or seedling growth of L. olgensis. More than 30% of the ground surface was not covered by any plants, showing that herbaceous and moss layers did not inhibit seedling emergence. The crown gaps were also more than 30% of the canopy. Therefore, the reason that there were no saplings less than 20 years old in the surveyed plot may be explained by the rarity of occurrence of proper regeneration conditions. This is seen in alder forests on swamps in a northern part of Japan (Yabe, 1989). The longevity of larch is generally short but is, at least, more than 50 years (Tatewaki, et al., 1965), showing that the surveyed forest is young. The lack of old stems may be due to the rarity of proper regeneration conditions. For the closely related Larix laricina the best seedbed is considered moist mineral or organic soil (Beeftink, 1955) a condition often created by flooding in wetlands (Yabe, 1989). Therefore, disturbances such as flooding may play an important role in the regeneration of L. olgensis. A large-scale drainage project is under construction in this peatland to improve soil conditions for agriculture. Seedling distribution patterns are sensitive to even microenvironmental changes in wetlands (Titus, 1990). This project, which would alter the flood regime and change water and soil conditions, will probably deal the L. olgensis forest a fatal blow. Therefore, the proper conservation and management of this L. olgensis forest is of immediately necessity. 
ACKNOWLEDGMENTSI thank all members of the Overseas Survey on Chinese Peatland for their help, and J.H. Titus and V.A. Nuzzo for critical readings of the manuscript. |