(Upload on September 11 2016) [ 日本語 | English ]
Mount Usu / Sarobetsu post-mined peatland
From left: Crater basin in 1986 and 2006. Cottongrass / Daylily
HOME > Lecture catalog / Research summary > Glossary > Virus
|
1892 Iwanovsky WP (USSR): discovering that tobacco mosaic virus (TMV) was filterable 1898 Beijerinck: porposed virus (L.) 1935 Stanley M 1904-1971, USA
TMV = globulin-like protein (corrected later → protein + nucleic acid) 1939 Pfankuck et al.: emphasized the importance of morphology (phenotypic classification) after the detailed observation of TMV 1953, 1957 Lwoff: summarized the characteristics of virus
1) present either DNA or RNA Virus groups based on the hostsBacterial virusinvertebrate virus Vertebrate virus Plant virus |
[classification, structure, biology] 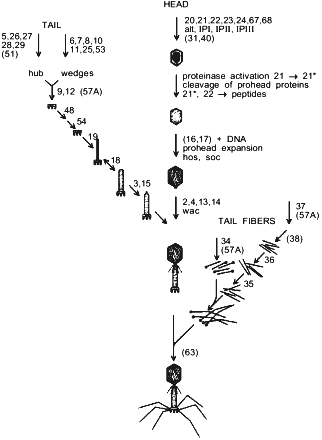
Pathway of bacteriophage T4 morphogenesis. Genes participating in assembly are numbered. Chaperones that are not part of the final structure are presented in parentheses. |
| Characterization | Families or groups | Kinds of host | |
| dsDNA, enveloped | Poxviridae Herpesviridae Baculoviridae Plasmaviridae | V, I V I bacteria | |
| dsDNA, non-enveloped | tailed phages | Iridoviridae * Adenoviridae Papovaviridae Caulimovirus Tectiviridae Corticoviridae Myoviridae Styloviridae Podoviridae | V, I V V P bacteria bacteria bacteria bacteria bacteria |
| ssDNA, non-enveloped | Parvoviridae Geminivirus Microviridae Inoviridae | V, I P bacteria bacteria | |
| dsRNA, enveloped | Cystoviridae | bacteria | |
| dsRNA, non-enveloped | possible family possible family possible family possible family | Reoviridae Isometric dsRNA mycoviruses requiring only one RNA segment for replication Isometric dsRNA mycoviruses requiring two RNA segments for replication Isometric dsRNA mycoviruses requiring three RNA segments for replication Bisegmented dsRNA viruses | V, I, P fungi fungi fungi V, I |
| ssRNA, enveloped Genome strategy | a. No DNA step (i) positive-sense genome (ii) negative-sense genome (iii) not established b. DNA step in replication cycle Bipartite genomes Isometric particles Possible group Rod-shaped particles Tripartite genomes Isometric particles Isometric and bacilliform particles Rod-shaped particles | Togaviridae Coronaviridae Paramyxoviridae Orthomyxoviridae Rhabdoviridae Bunyaviridae Arenaviridae Tomato spotted wilt virus group Retroviridae Dianthovirus Comovirus Nepovirus Pea enation mosaic virus group Nodaviridae Velvet tobacco mottle virus group Tobravirus Cucumovirus Bromovirus Ilarvirus Alfalfa mosaic virus group Hordeivirus | V, I V V V V, I, P V, I V P V P P P P I P P P P P P P |
| ssRNA, non-enveloped | Monopartite genomes Isometric particles Rod-shaped particles | Picornaviridae Caliciviridae Nudaurelia β virus group Leviviridae Maize chlorotic dwarf virus group Tymovirus Luteovirus Tombusvirus Sobemovirus Tobacco necrosis virus group Closterovirus Carlavirus Potyvirus Potexvirus Tobamovirus | PV, I V I bacteria P P P P P P P P P P P |
ICTV (International Committee on Taxonomy of Viruses) (2015)
Virus families not assigned to an OrdFam AdenoviridaeFam AlphatetraviridaeFam AlvernaviridaeFam AmalgaviridaeFam AmpullaviridaeFam AnelloviridaeFam ArenaviridaeFam AscoviridaeFam AsfarviridaeFam AstroviridaeFam AvsunviroidaeFam BaculoviridaeFam BarnaviridaeFam BenyviridaeFam BicaudaviridaeFam BidnaviridaeFam BirnaviridaeFam BromoviridaeFam BunyaviridaeHantaviruses: single-stranded, enveloped, negative sense RNA viruses that can kill humans Fam CaliciviridaeFam CarmotetraviridaeFam CaulimoviridaeFam ChrysoviridaeFam CircoviridaeFam Clavaviridae |
Fam ClosteroviridaeFam CorticoviridaeFam CystoviridaeFam EndornaviridaeFam FlaviviridaeFam FuselloviridaeFam GeminiviridaeFam GenomoviridaeFam GlobuloviridaeFam GuttaviridaeFam HepadnaviridaeFam HepeviridaeFam HypoviridaeFam HytrosaviridaeFam InoviridaeFam IridoviridaeFam LavidaviridaeFam LeviviridaeFam LuteoviridaeFam MarseilleviridaeFam MegabirnaviridaeFam MetaviridaeFam MicroviridaeFam MimiviridaeFam NanoviridaeFam NarnaviridaeFam NimaviridaeFam NodaviridaeFam Nudiviridae |
Fam OphioviridaeFam OrthomyxoviridaeFam PapillomaviridaeFam PartitiviridaeFam ParvoviridaeFam PermutotetraviridaeFam PhycodnaviridaeFam PicobirnaviridaeFam PlasmaviridaeFam PleolipoviridaeFam PolydnaviridaeFam PolyomaviridaeFam PospiviroidaeFam PotyviridaeFam PoxviridaeFam PseudoviridaeFam QuadriviridaeFam ReoviridaeFam RetroviridaeFam SarthroviridaeFam SphaerolipoviridaeFam SpiraviridaeFam TectiviridaeFam TogaviridaeFam TombusviridaeFam TotiviridaeFam TurriviridaeFam UnassignedFam VirgaviridaeICTV (2015) |
|
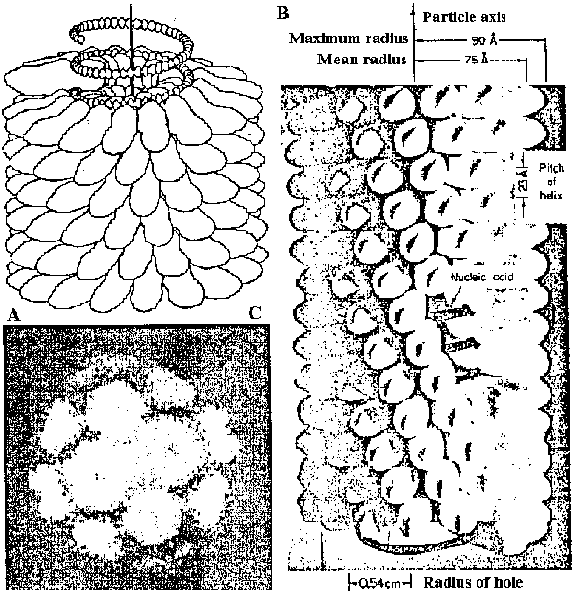
TMV (タバコモザイクウィルス)300 nm in height × 18 nm in widthrod-shaped virus → applicable this basic structure to rod-shaped and string-shaped viruses present 2130-17500 dt of proteins components: protein = 95% + RNA 5% Fig. Structure of TMV. (A) Drawing to show the relationships of the RNA and the protein subunits. The DNA shown free of protein could not maintain this configuration in the absence of the protein subunits. There arc 3 nucleotides per protein subunit, or 49 per turn of the major helix spaced about 5 A apart. (B) Photograph of a model of TMV with major dimensions indicated. The structure repeats after 69 A in the axial direction, the repeat containing 49 subunits distributed over 3 turns of the helix.(Klug & Caspar 1960, and based largely on the work of R. E. Franklin) (C) Photograph of a particle of TYMV obtained by symmetrizing an electron microgrpah of a single particle. (Markham 1968) The molecular weights of basic proteins differ between this TMV and the other TMV. Many species have proteins of which molecular weights are approximately 18-Kd → attempting to apply virus classification |
|
RhabdovirusEnveloped, bullet shaped. 180 nm long and 75 nm wide. Certain plant rhabdoviruses are bacilliform in shape and almost twice the length. The basic structure is comparable with the structure of TMV.→ Viron |
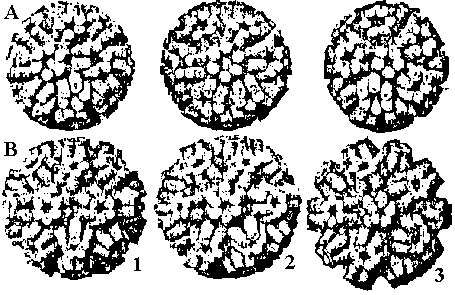
Spherical virus (球形ウィルス)All spherical virus shells have Icosahedral symmetry = Built from 20 identical equilateral triangles.→ enclose maximum volume → probably structurally most stable The structures were predicted by of subunit sequences 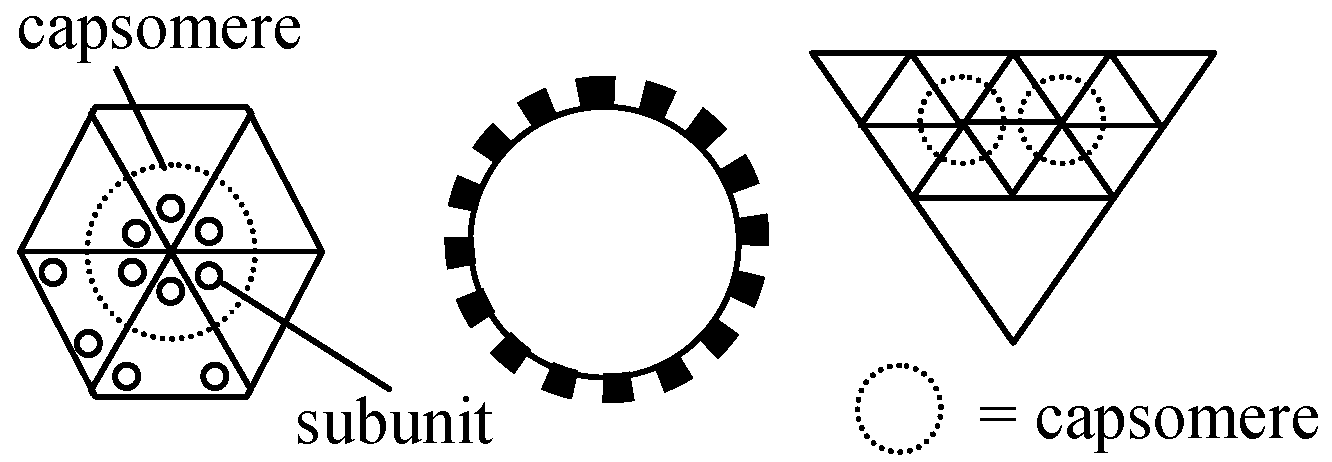
The surface structures of spherical virus is estimated by the variations of the combinations of subunits and/or the relationships between variations of subunit shapes and numbers of subunits Fig. A. Comparison of models each built of 180 units (corks) on the surface of a sphere representing in position, direction and radial extent the structure units in the outer regions of thc protein shells of A BBMV, B TYMV (Finch & King 1966). 1) Twofold view, 2) threefold view, 3) fivefold view. The structure units of BBMV protrude ca 15 A from the body of the particle and lie with their long axes approximately parallel to the nearest local sixfold or fivefold axis of the particle, forming 32 cylindrical or prismatic morphological units. The structure units of TYMV protrude further from the body of the particle than do those of BBMV and are tilted further toward the nearest local sixfold or fivefold axis to form morphological tin its shaped like frusta of cones. (Finch & King 1967) Fig. B. Clustering patterns of subunits in icosahedra. Top left is a simple icosahedron. Its triangular faces are undivided (T = 1): each is defined by three subunits and thus there are 60 subunits in the entire icosahedron. These subunits may cluster in fives (a, pentamer clusters) at the vertices as in the particles of satellite virus. Middle left is an icosadeltahedron of 180 subunits. Each triangular face of the underlying icosahedron (broken lines) is divided into six half triangles (unbroken lines: T = 6 × 1/2 = 3). Each triangular face is defined by three subunits, and these could cluster together (trimers), or may cluster with subunits of adjacent triangles either in pairs (dimers), or in fives and sixes (pentamer/hexamers). Dimer clustering gives, in one view, a 'Star of David' pattern (b), and this is found in particles of turnip crinkle virus. Pentamer/hexamer clustering (d) is seen in particles of tymoviruses. The particles of some viruses, such as alfalfa mosaic virus are tubular variants of an icosadeltahedron (c). These particles consist of two half icosadeltahedra joined by a tubular net, which has no five-fold axes. The clustering pattern in alfalfa mosaic virus particles is not known for sure; the subunits may be clustered as pentamer/hexamers (c. top half) or trimers (c. lower half). |
 A A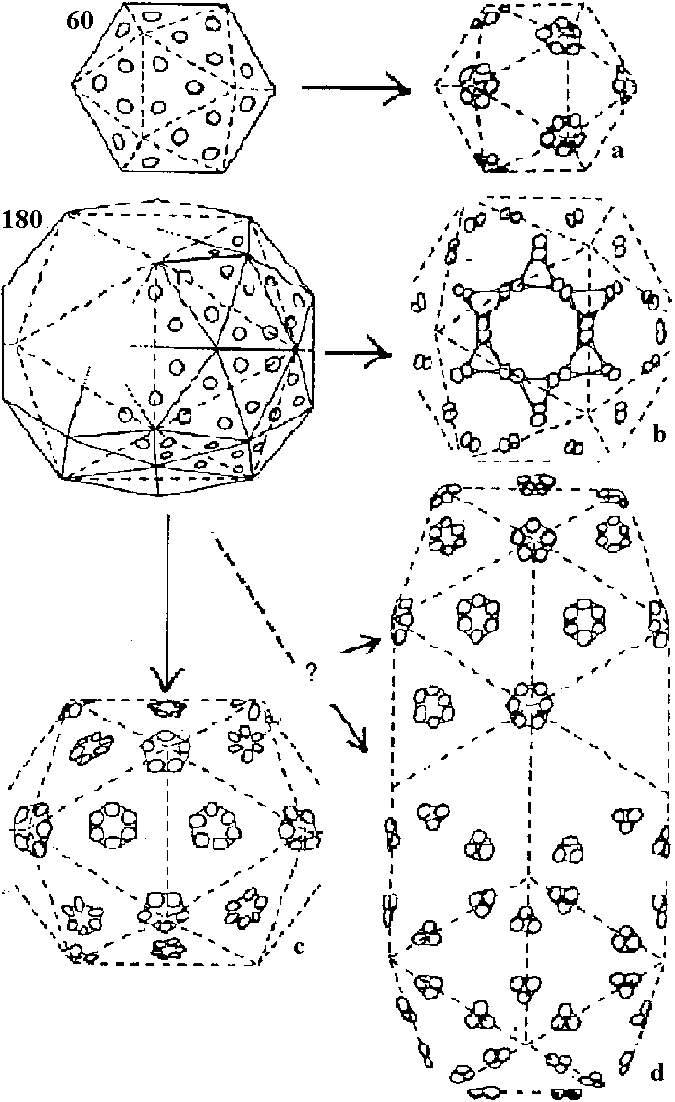 B B
|
Multicomponent and segmental genomeOnly seen in plant virusInformation expression mechanisms on genome or gene genome (= nucleic acids) → enzyme, protein (coat protein) = form, etc.
TMV = 5% nucleic acids + 95% proteins |
| Group | NL (rnp) | Shape | φ (μm) | Transmission | TIP- (°C) | a | b | |
|---|---|---|---|---|---|---|---|---|
| 1 | 130 | Rigid rods | 20 | Sap; soil | - | Barley stripe mosaic virus (1) | Soil-borne wheat mosaic virus (2) | |
| 2 | 180 | Rigid rods | 75-80 | Potato stem mottle virus (3) | ||||
| 3 | 300 | Rigid rods | 15 | Sap | 85-95 | + Tobacco mosaic virus (4) + Cowpea virus (5) | + Cucumber green mottle virus (6) Odonioglossum ring spot virus (7) | |
| 4 | 480 | Flexible threads | 10-11 | Sap; (aphids?) | 60-75 | White clover mosaic virus (8) | Cymbidium mosaic virus (7) | |
| 5 | 515 | Flexible threads | Potato virus X (9) | Cactus virus 1 (10) | ||||
| 6 | 580 | Flexible threads | Potato aucuba mosaic virus (11) | |||||
| 7 | 620 | Rods, rigid to slightly flexible | 12-13 | Apids; sap | 65-75 | Wisconsin pea stieak virus (12) | ||
| 8 | 650 | Rods, rigid to slightly flexible | Red clover vein mosaic virus (12) * Carnation latent virus (13) * Potato virus S (13) * Potato virus M (13) Cactus virus 2 (10) | "Hop virus" (14) "Gladiolus virus"(14) "Poplar virus" (14) Tulip-breaking virus (15) | ||||
| 9 | 700 | Rods, rigid to slightly flexible | 12-13 | Mites; sap | - | Wheat streak mosaic virus (16) | ||
| 10 | 730 | Flexible threads | 12-13 | Aphids; sap | 50-60 | Beet mosaic virus (17) Potato virus A (18) Potato virus Y (18) Tobacco etch virus (19) Henbane mosaic virus (19) | ||
| 11 | 750 | Flexible threads | ‡ Bean common mosaic virus (20) ‡ Bean yellow mosaic virus (20) ‡ Pea mosaic virus (20) Soybean niosaic virus (20) Turnip mosaic virus (21) Cockaf not streak virus (19) Lettuce aulusaic virus (22) Sorghum red stripe virus (23) | Sugar cane mosaic virus (24) | ||||
| 12 | 1250 | Very flexible threads | 10 | Aphids; (sap) | - | Beet yellows virus (25) |
a, b, +, *, ‡ Indicate serological relationship within groups 3, 8, and 11, respectively. Numbers in parentheses refer to footnotes as follows: (1) Gold et al., 1954: 130 × 30 μm. Brandes (1959) gave a NL of 128 μm, but the diameter of the particles was only about 20 μm when compared with TMV and potato stem mottle virus. (2) Gold et al., 1957. (3) Paul & Bode (1955) found two frequent lengths: 70 and 180 μm. The values of Harrison & Nixon (pers comm) are in good accordance. These authors could show that the shorter particles are noninfectious. Therefore, 180 μm must be taken as NL. Potato stem mottle virus also includes tobacco rattle virus and soil-borne potato viruses found in the United States (Walkinshaw & Larson 1958, Oswald & Bowman 1958). The morphological data are apparently the same; furthermore, serological relationship has been established. (4) A great number of papers exist; 300 μm is especially the result of detailed measurements by Williams & Steers (1951). TMV often has been used as a standard in our laboratory. (5) The cowpea virus was kindly supplied by FC Bawden. (6) A detailed study on this subject is that of Knight (1955). (7) Jensen & Gold, 1951; Newton & Rosberg, 1952; Gold & Jensen, 1953; Murakishi, 1958. (8) Brandes & Quanta, 1957. (9) Bode & Paul, 1955. (10) With the aid of the dipping method we found two different viruses in several species of cactus plants (Brandes & Uachdraweit, unpublished). Cactus virus 1 possibly is identical with a virus described by Amnelunxen (1958). (11) Paul & Bode, 1956. (12) Wetter et al., 1959; Wetter & Quanta, 1953; Brandes & Quanta, 1957. (13) Brandes et al., 1959. (14) Electron microscopic studies of these viruses are going on in our laboratory. The hop virus is being studied by Nuber K (Landwirtschaftliche Hochschule, Hohenheim). (15) De Bruyn Ouboter et al., 1951. (16) Gold et al., 1957; Brandes, 1959. (17) Zimmer & Brandes, 1956. (18) Bode & Paul, 1956; Paul & Bode, 1956; Brandes & Paul, 1957. (19) Brandes, 1959. (20) These four legume viruses all have the same size (Brandes & Quanta 1955, Quanta 1959). Serological relationship has been established between bean common mosaic virus and bean yellow mosaic virus (Beenister & van der Want, 1951; Bereks, pers comm) and between bean yellow mosaic virus and pea mosaic virus (Goodchild. 1956). (21) Bode & Brandes, 1958. (22) Couch & Gold, 1954: 748 × 22 mm. Our determination of the NL revealed the same value (747 mm), but measurements in comnparison with TMV and other viruses revealed a diameter of about 12-13 μm (Brandes 1959). (23) This virus has been described and suspected of being related to sugar cane mosaic virus by Grancini (1957). It has been bee measured in our laboratory (Brandes 1959). (24) Gold et al., 1957. (25) Brandes & Zimnmer, 1955; Burghardt & Brandes, 1957.
| Name | Vector | Geographical distribution | In situ distribution | S | Nucleic acid | SCPS | |
| Type | Size | ||||||
| Maize streak | Cicadulina rnbila | Africa, India | - | 76 | ssDNA | 7.1 × 105 | 28000 |
| Bean golden mosaic | Bemisia lubaci | Tropical Am. | Phloem-associated parenchyma & sieve elements | 69 | ssDNA | 8.0 × 105 | 27400 |
| Cassava latent | - | Africa | - | 76 | ssDNA | 8.0 × 105 | 34000 |
| Chloris striate mosaic | Nesoclutha pallida | Australia | General but not epidermis | - | ssDNA | 7.1 × l05 | 28000 |
| Curly top | Circulifer tenellus | North Am. | Phloem-associated parenchyma | - | - | ||
| Euphorbia mosaic | Bemisia tabaci | Tropical Am. | Phloem-limited | - | - | ||
| Tomato golden mosaic | Bemisia tabaci | Tropical Am. | Phloem-limited | - | - | ||
| Tomato yellow dwarf | Bemisia tabaci | Japan | Phloem-limited | - | - | ||
| Tobacco leaf curl | Bernisia tabaci | Pantropical | - | - | - | ||
| Tobacco yellow dwarf | Orosius argentatus | Australia | - | 76 | - | 27500 | |
| Bean summer death | Orosuis argentatus | Australia | Phloem-limited | - | - | - | |
| Mungbean yellow mosaic | Bemisia tabaci | South Asia | - | - | - |
| # | WTV | RDV | FDV | RBSDV | MRDV | PSV | ||||||||||||
| PP | PP | PP | ||||||||||||||||
| R | MWcal | MWobs | No | Loc | R | MWcal | MWobs | No | Loc | R | R | R | MWcal | MWobs | No | Loc | R | |
| 1 | 2.90 | 170.6 | 160.0 | I | C | 3.10 | 194.0 | 193.0 | I | C | 2.90 | 2.91 | 2.88 | 160.0 | ||||
| 2 | 2.40 | 141.2 | 131.0 | II | OL | 2.50 | 156.0 | 152.0 | II | OL | 2.50 | 2.50 | 2.50 | 138.0 | 139.0 | I | C | 2.54 |
| 3 | 2.20 | 129.4 | 118.0 | III | C | 2.20 | 137.5 | 131.0 | III | C | 2.48 | 2.35 | 2.35 | 130.0 | 126.0 | II | C | 2.50 |
| 4 | 1.80 | 105.9 | 1.80 | 112.5 | 2.48 | 2.35 | 2.35 | 130.0 | 123.0 | III | B | 2.50 | ||||||
| 5 | 1.78 | 104.7 | 96.0 | IV | OL | 1.76 | 110.0 | 110.0 | IV | OL | 2.12 | 2.12 | 2.12 | 117.0 | 111.0 | IV | OC | 2.12 |
| 6 | 1.10 | 64.7 | 1.05 | 65.6 | 1.85 | 1.75 | 1.75 | 97.0 | 97.0 | V | OC | 1.75 | ||||||
| 7 | 1.05 | 61.8 | 58.0 | V | C | 1.02 | 63.7 | 62.0 | V | C | 1.45 | 1.45 | 1.45 | 80.0 | 1.48 | |||
| 8 | 0.83 | 48.8 | 36.0 | VI | Ca | 0.78 | 48.8 | 46.0 | VI | OC | 1.21 | 1.25 | 1.25 | 68.0 | (1.25) | |||
| 9 | 0.57 | 33.5 | 35.0 | VII | Ca | 0.70 | 43.8 | 45.0 | VII | OC | 1.15 | 1.18 | 1.18 | 65.0 | 64.0 | VI | OC | |
| 10 | 0.55 | 32.4 | 0.67 | 41.9 | 1.12 | 1.10 | 1.08 | 60.0 | ||||||||||
| 11 | 0.54 | 31.7 | 0.48 | 30.0 | ||||||||||||||
| 12 | 0.32 | 18.8 | 0.48 | 30.0 | ||||||||||||||
| Total | 16.0 | 16.6 | 19.3 | 19.0 | 18.9 | (19.6) |
1. Contact (接触)2. Seed transmission (種子)3. Soil transmission (土壌)a. Nematode (線虫)Transmission of nepovirus and tobravirus through nematode
Group cryptogram: [R/1:2.4/42/(1.4 – 2.2)/(27 – 40) – (2 × 1.4)/46:S/S:S/C, Ve/Ne RNA-1 a. RASPBERRY RINGSPOT VIRUS
Seed transmissibility in Stellaria media
Seed transmissibility in S. media
? RNA polymerase production a. RASPBERRY RINGSPOT VIRUS
Coat protein specificity
Coat protein specificity |
c. TOBACCO RATTLE VIRUS
Coat protein specificity b. Fungi (菌類)4. Sap transmission (汁液)5. Insect transmission (虫媒)Aphid (アブラムシ)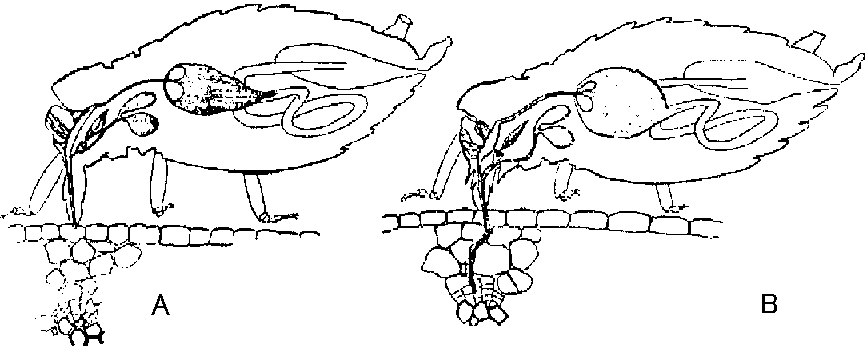 Fig. Illustration of aphids in probing position. (A) Aphids on position to probe virus-infected tissues of the epidermis. (B) Aphids in feeding position, acquiring virus from phloem cells. Leaf beetle (ハムシ)= chrysomelidTick (ダニ)Whitefly (コナジラミ)Issues1. genome maskingTable 6. General pattern of transmission (+) or non-transmission (-) of 5 isolates of barley yellow dwarf virus by 4 aphid species. Virus isolates R. padi M. avenae R. maidis S. graminum PAV + + - + RPV + - - + MAV - + - - RMV - - + - SGV - - - +→ a mechanism that the early-infected virus protects against the infection of phylogenetically-different virus ↔ this virus does not have this mechanims = simultaneous replication 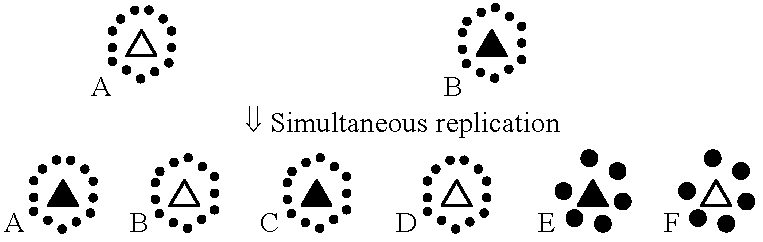 Fig. 6. The combinations of proteins and nucleic acids extracted from the strains concomitantly infected by two virus species. C, D: transcapsidation, E, F: phenotypic mixing. 2. helper component |
|
The smallest known pathogens, are naked, circular, single-stranded RNA molecules (200-400 bases) that do not encode protein yet replicate autonomously when infected into host vascular plants 1971 Diener, Theodor Otto (1921-2023, Swiss-American)
discovered potato spindle tuber viroid (PSTVd, ジャガイモやせ芋ウイロイド) = 130000 Da TaxonomyNomenclature: described in English (not Latin)host plant + characterized desease + viroid Similarity of genome sequence > 90% ⇒ same speciesvarieteis when needed, even if siilarity > 90% ≥ 30 viroidsFamily Avsunviroidae Avsunviroid: Avocado sunblotch viroid (ASBVd) Pelamoviroid: Chrysanthemum chlorotic mottle viroid (キク退緑斑紋ウイロイド), Peach latent mosaic viroid (モモ潜在モザイクウイロイド) Elaviroid: Eggplant latent viroid |
Family Pospiviroidae Pospiviroid: Chrysanthemum stunt viroid (キク矮化ウイロイド), Citrus exocortis viroid (カンキツエクソコーティスウイロイド), Columnea latent viroid, Iresine viroid 1, Mexican papita viorid, Pepper chat fruit viroid, Potato spindle tuber viroid, Tomato apical stunt viorid, Tomato chlorotic dwarf viroid (トマト退緑萎縮ウイロイド), Tomato planta macho viroid Hostuviroid: Hop stunt viroid (ホップ矮化ウイロイド) Cocadviroid: Citrus bark cracking viroid (カンキツバーククラッキングウイロイド), Coconut cadang-cadang viroid, Coconut tinangaja viroid, Hop latent viroid (ホップ潜在ウイロイド) Apscaviroid: Apple dimple fruit viroid (リンゴくぼみ果ウイロイド), Apple scar skin viroid (リンゴさび果ウイロイド), Australian grapevine viroid (ブドウオーストラリアウイロイド), Citrus bent leaf viroid (カンキツベントリーフウイロイド), Citrus dwarfing viroid (カンキツ矮化ウイロイド), Citrus viroid V (カンキツウイロイドV), Citrus viroid VI (カンキツウイロイドVI), Grapevine yellow speckle viroid 1 (ブドウ黄色斑点ウイロイド1), Grapevine yellow speckle viroid 2, Pear blister canker viroid (ナシブリスタキャンカーウイロイド) Coleviroid: Coleus blumei viroid 1 (コリウスウイロイド1), Coleus blumei viroid 2, Coleus blumei viroid 3 Disease symptom of hostStem and leaf: mottle, stunt, dwarf and yellow spotWoody plants: mottle, epinasty (葉下垂), exocortis (樹皮剥離) |