(Upload on May 1 2026) [ 日本語 | English ]
Mount Usu / Sarobetsu post-mined peatland
From left: Crater basin in 1986 and 2006. Cottongrass / Daylily
HOME > Lecture catalog / Research summary > Glossary > Enzyme
Chemical change (化学変化)Chemical equation (化学反応式) → chemical property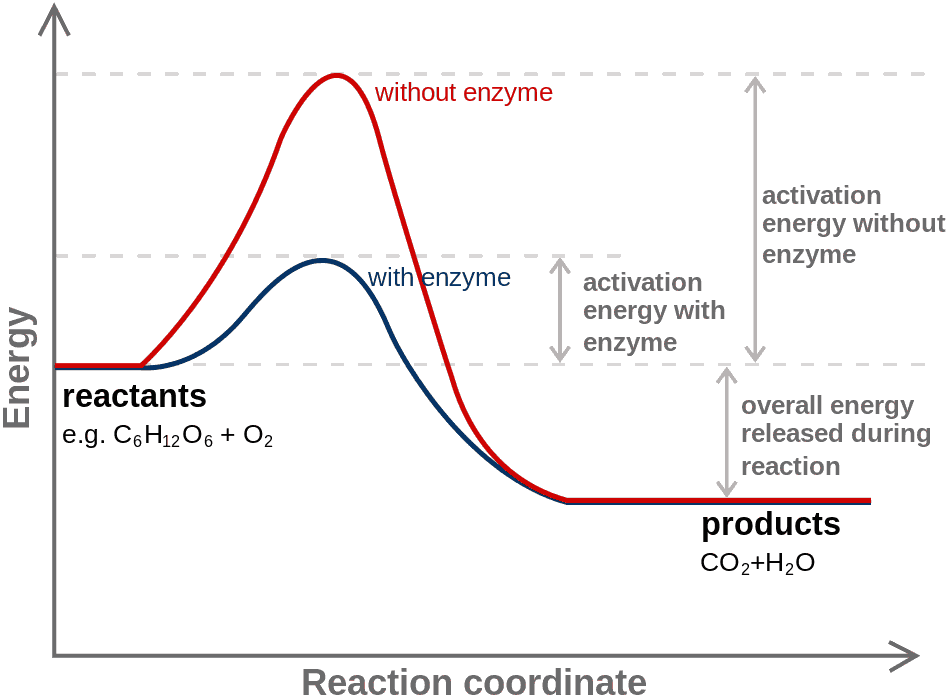 The energy variation as a function of reaction coordinate shows the stabilisation of the transition state by an enzyme. Chemical equilibrium (化学平衡)condition in the course of a reversible chemical reaction in which no net change in the amounts of reactants and products occurschemically stable ⇔ chemically unstable |
[ catalyst | enzymology | kinetics ]
|
|
____MnO2 (catalyst) H2O2 → 2H2O + O2 _____MnO2 (catalyst) 2KClO3 → 2KCl + 3O2 |
Enzyme (酵素)0. effective with infinitesimal1. catalyst
A → [catalyst or enzyme] → P
To promote reaction |
2. temperature effect 3. pH effect (v-pH relationship) 4. specificity |
[ vitamin ]
Holoenzyme= apoenzyme + coenzymeEx. heme (containing Fe) + protein = cytochromes Flavin (フラビン)Isoalloxazine ring - basic skeleton of flavin
FAD (flavin adenine dinucleotide): attached with an adenosine diphosphate
AH2 + F ↔ A + FH2
NAD·NADP (pyridine nucleotides) (NAD·NADP)Thiamin (Thiamine, チアミン)thiazole ring
|
Pyridoxine (ピリドキシン)Pyridoxial-P-α-AA → Pyridoxiamine-P + α-keto acidBiotin (vitamin B7, ビオチン)= formerly called vitamin H or coenzyme R1916 Bateman: a raw egg white diet caused toxicity in various species 1927 Boas & Parsons: discovered a yeast growth enhancer - named bios 1931 György: called vitamin H, from Haut (skin) in German 1936 Kögl: isolated the enhancer from yolk 1941 György: egg-white injury caused by biotin deficiency excessive consumption of raw egg whites → (cooked egg whites do not cause the problems)
→ biotin binds strongly to avidin in raw egg whites → Coenzyme A (CoA, 補酵素A)the synthesis and oxidation of fatty acidsthe oxidation of pyruvate in the citric acid cycle
Ubiquinone, CoQ, UQ (ユビキノン)Summary of coenzymesEnzyme: Type of covalent intermediate
|
Metalslow molecular and stable to heatZn2+: alcohol dehydrogenase, carbonic anhydrase, carbonicpeptidase Mg2+: phosphohydrolases, phosphotransferases Mn2+: arginase, phsphotransferases Fe2+ or Fe3+: cytochromes, peroxidase, catalase, ferredoxin Cu2+ or Cu+: tryosinase, cytochrome oxydase Mo: nitrogenase K+: pyruvate kinase (also required Mg2+) Na+: plasma membrane ATPase (also required K+ and Mg2+) |
Flavin mononucleotide、FMN= riboflavin-5'-phosphate (riboflavin = vitamin B2), a phosphorylated form of riboflavinassisting NADH dehydrogenase |
Zero-order reaction (0次反応)First-order reaction (1次反応) |
Inhibition patternreversible inhibition = competitive inhibition + noncompetitive inhibitionirreversible inhibition Reversible inhibition (可逆阻害)Competitive inhibition (拮抗阻害)interruption of a chemical pathway owing to a chemical substance inhibiting the effect of another by competing with it for binding or bonding1/V0 = (1 + 1/K1)Km/Vmax·1/[S] + 1/Vmax
K1 = [E][I]/[EI] Noncompetitive inhibition (非拮抗阻害)Irreversible inhibition (非可逆阻害) |
Table. Summary of reversible enzyme inhibition Vappmax Vappmax/KappM KappM competitive (拮抗阻害): KM↑ Vmax (unaffected)Vmax (Vmax/KM)/(1 + [I]/KI) KM(1 + [I]/KI) mixed (混合阻害)Vmax/(1 + [I]/KIu) (Vmax/KM)/(1 + [I]/KI) KM{(1 + [I]/KI)/(1 + [I]/KIu)} noncompetitive (非拮抗阻害): KM (unaffected) Vmax↓Vmax/(1 + [I]/KI) (Vmax/KM)/(1 + [I]/KI) KM uncompetitive (不拮抗阻害): KM↓ Vmax↓Vmax/(1 + [I]/KIu) Vmax/KM KM(1 + [I]/KIu) ⇓ Lineweaver-Burk plot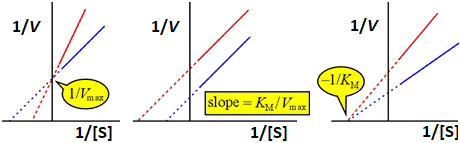 Competitive Uncompetitive Noncompetitive inhibition inhibition inhibition ━━━┅┅┅ inhibitor. ━━━┅┅┅ no inhibitor |
Digestive enzyme (消化酵素) |
Enzyme Committee, EC (酵素委員会)Recommendations of the Nomenclature Committee of the International Union of Biochemistry and Molecular Biology on the Nomenclature and Classification of Enzymes by the Reactions they CatalyseEvery enzyme code consists of the letters "EC" followed by four numbers separated by periods Those numbers represent a progressively finer classification of the enzyme. Preliminary EC numbers exist and have an 'n' as part of the fourth (serial) digit (e.g. EC 3.5.1.n3). Three names of enzymesTrivial name (常用名), called in common useRecommended name (推奨名), recommended by the newest admonishment from EC Systematic name (系統名) Classification proposed by EC1961 International Union of Biochemistry (in Moscow)
establishing the officially accepted rule of enzyme names
|
1. = Oxidoreductases 1961 (yr) = 712, 1964 = 875, 1972 = 1770, 1975 = 1974, 1979 = 2122, present > 3000 |