(Upload on December 14 2020) [ 日本語 | English ]
Mount Usu / Sarobetsu post-mined peatland
From left: Crater basin in 1986 and 2006. Cottongrass / Daylily
HOME > Lecture catalog / Research summary > Glossary > Chemistry > Chemicals
[ プロトコル ]
|
Chemistry: divided into two subjects based on chemical compounds
= organic chemistry (有機化学) + inorganic chemistry (無機化学) Organic chemistryresearch on the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms (except simple carbon compounds)
Natural products chemistry (天然物化学): the study of chemical compounds produced by living organisms
focusing on isolation, structural elucidation, biosynthesis, properties, and functions |
Inorganic chemistryresearch on the synthesis and behavior of inorganic and organometallic compounds (i.e., non-organic compounds)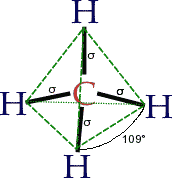
|
|
mole number or molar number |
Ionic compounds (イオン化合物)NO2-: nitrite, NO3-: nitrateSO32-: sulfite, SO42-: sulfate, HSO4-: hydrogen sulfate ion (/bisulfate) (bi = H) CO32-: carbonate, HCO3-: hydrogen carbonate (/bicarbonate) PO43-: phosphate, HPO42- :hydrogen, H2PO4-; dihydrogen (di + 2 atoms) ClO-: hypochlorite (less oxygen), ClO2-: chlorite, ClO3-: chlorate, ClO4-: perchlorate (more oxygen) |
|
alkyl groups aryl groups |
Ether (エーテル)R-O-R'__R, R' = alkyl or aryl groups
Alcohol (アルコール)Ethanol (エタノール) Phenol (フェノール)Aldehyde and ketone (アルデヒド/ケトン)Carboxylic acid (カルボン酸)Ester (エステル) |
Redox (oxidation-reduction or oxidoreduction, 酸化還元)Def. oxidation-reduction potential (ORP) (酸化還元電位)= redox potential, or electrode potential on the hydrogen scale (Eh) Redox indicator (酸化還元指示薬)= oxidation-reduction indicatorindicators that undergo a color change at a specific electrode potential fast and reversible color change Ex. KMnO4
• metal complexes of phenanthroline and bipyridine 2, 3, 5-triphenyl tetrazolium chloride (TTC), C19H15ClN4
Also used for checking seed viability
|
Table. Major reducing substances and reduction systems reacted to tetrazolium salts
Electrolysis (電気分解)
Platinum electrode (白金電極) |
|
Saturated straight chain (飽和直鎖状) Saturated branched (飽和分枝状) Saturated chain (飽和環状) (cycloparaffine 飽和環状炭化水素) Unsaturated straight chain (不飽和直鎖状) Unsaturated chain (不飽和環状) |
= carbohydrateClassificationAlkane (アルカン)CnH2n + 2 → IUPAC nomenclature
n Eq. nom. B/Mp AOTP Isomers SSF
1 CH4 Methane -162 /-183 gas 0 CH4
2 C2H6 Ethane -89 /-184 gas 0 CH3-CH3
3 C3H8 Propane -42 /-187 gas 0 CH3-CH2-CH3
4 C4H10 n-butane -0.5/-138 gas 2 CH3-CH2-CH2-CH3
i-butane CH3-CH3-CH-CH3
5 C5H12 n-pentane 36 /-130 liquid 3
iso-pentane
neo-pentane
6 C6H14 n-hexane 69/-95 liquid 5
iso-hexane
…
10 C10H22 liquid 95
14 C14H30 liquid 1858
→ many isomers → limitations of nomenclature |
Eq.: equation, nom.: nomenclature, B/Mp (oC): boiling and melting points, AOTP: at ordinary temperatures and pressures, SSF: shortened structural formula |
Monosaccharide (単糖類)Uronic acids (ウロン酸)Fucose (フコース)C6H12O5 L-rhamanose (6-deoxy-L-mannose)L-fucose (6-deoxy-2-galactose) Pentose (五炭糖) Arabinose
L-phamnose (6-deoxy-L-mannose) L-fucose (6-deoxy-L-galactose) Glucose (グルコース, ブドウ糖)
CHO
glyceraldehyde(basic molecule) |
Starch (デンプン)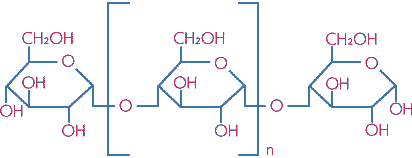 |
|
Succinic acid (コハク酸): HOOC-CH2-CH2-COOH Malic acid (リンゴ酸): HOOC-CH2-CH(OH)-COOH Fumaric acid (フマル酸): HOOC-CH=CH-COOH Oxaloacetate (オキザロ酢酸): HOOC-CH2-CO-COOH Citric acid (クエン酸): C(OH)(CH2COOH)2COOH
|
Pyruvic acid (ピルビン酸)CH3COCOOH
= 2-oxopropanoic acid (2-オキソプロパン酸), IUPAC |
|
Biologically important organic compounds containing amine (-NH2) and carboxylic acid (-COOH) functional groups carbon on the center: asymmetric carbon → carboxylic radical Natural amino acids (天然アミノ酸)1) Non-polar amino acid (非極性アミノ酸)R =1) -H: glycine (グリシン, Gly, G) 2) -CH3: alanine (アラニン, Ala, A) 3) -CH2(CH3)-CH·CH3: leusine (ロイシン, Leu, L) 4) -CH(CH3)-CH2·CH3: isoleusine (イソロイシン, Ile, I) 5) -CH2-S-CH3: methionine (メチオニン, Met, M) 6) -CH2- 7) 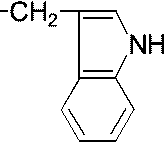 : tryptophane (トリプトファン, Trp, W) : tryptophane (トリプトファン, Trp, W)8) 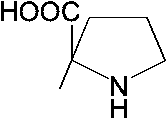 : proline (プロリン, Pro, P) : proline (プロリン, Pro, P)9) -CH(CH3)-CH3: valine (バリン, Val, V) 2) Polar and charge-free amino acids (極性だが電荷のないアミノ酸)10) -CH2OH: serine (セリン, Ser, S)11) -CHOH·CH3: threonine (スレオニン, Thr, T) 12) -CH2SH: cysteine (システィン, Cys, C) 13) -CH2- 14) -CH2CONH2: glutamine (グルタミン, Gln, Q) 15) -CH2CH2CONH2: asparagine (アスパラギン, Asn, N) |
3) Charged amino acids (電荷を持つアミノ酸)i) positively-charged amino acids16) -(CH2)4-NH2: lysine (リジン, Lys, K) 17) 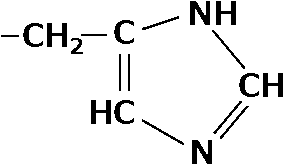 : histidine (ヒスチジン, His, H) : histidine (ヒスチジン, His, H)18) -CH(NH2)-CH-NH-C=NH: arginine (アルギニン, Arg, R) ii) negatively-charged amino acids 19) -CH2COOH: glutaminic acid (グルタミン酸, Glu, E) 20) -CH2CH2COOH: asparatic acid (アスパラギン酸, Asp, D) The proteins are formed by these 20 amino acids D-typed amino acids do not exsist in the nature 4) Non-proteinous amino acids (非タンパク質性アミノ酸)not present in proteins-(CH2)3-NH2: L-ornithine (L-オルニチン, Orn) -(CH2)3-NH(O=C-NH2): L-citrulline (L-シトルリン, Cit) → these two amino acids were discovered in ornithine cycle NH2-CH2-CH2-COOH: β-alanine (β-アラニン)Coenzyme A: a structural component of pantothenic acid (パントテン酸) 21) 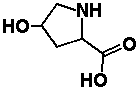 hydroxyproline (ヒドロキシプロリン) hydroxyproline (ヒドロキシプロリン)
→ occupying 12% of proteins in collagen 22) -(CH2)2-CH·OH-CH2-NH2: hydroxylisine (ヒドロキシリジン)→ rich in collagen, as well as hydroxyproline → number of essential amino acids (for the structural components of proteins) is 22 when these two amino acids are included |
|
Table. Classification of proteins based on biological functions __• Types and examples ___ function
|
|
|
polus (G. many or much) + phenol = polyhydroxyphenols: large multiples of phenol structural units Phenolic acid (フェノール酸)Rutin (ルチン)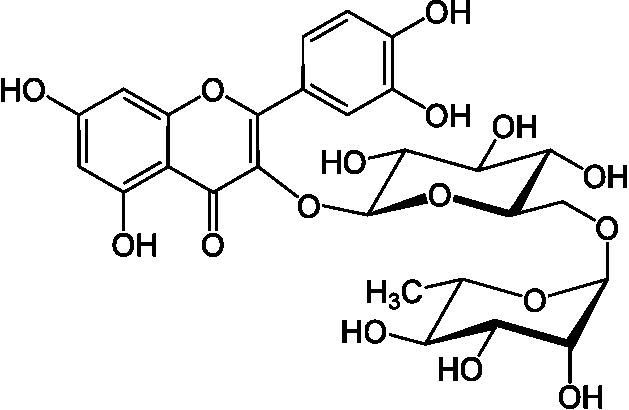
|
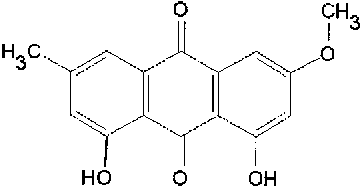
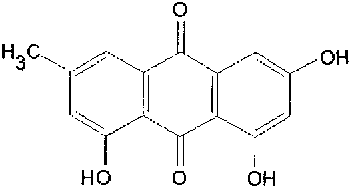 Anthraquinone physcion_____________Anthraquinone edmodin 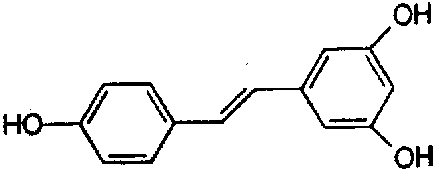 Resveratrol (レスベラトロール): Giant knotweed contains much resveratrol, one of the biologically active molecules. |
Sterol (ステロール)= steroid alcohol
Cholesterol (コレステロール)C27H46O: a sterol (or modified sterol) in lipids
a precursor for the biosynthesis of steroid hormones, bile acid and vitamin D |
Cholesteryl ester (コレステロールエステル), CE |