(Upload on January 31 2022) [ 日本語 | English ]
Mount Usu / Sarobetsu post-mined peatland
From left: Crater basin in 1986 and 2006. Cottongrass / Daylily
HOME > Lecture catalog / Research summary > Glossary > Seedbank
[ seedbank estimation | centrifuged flotation method ] [ seed | seed dispersal | seed germination ]
Seedbank, seed-bank, or seed bank (≈ buried seeds)
Seedbank dynamicsBasic equation (same with population dynamics)s = (p + i) - (c + c' + o), where
s: number of seeds present in a given soil c' = d + h (d: dead (s.s.), h: fed by herbivores) o: number of seeds escaped from the soil Seasonal fluctuations (季節変動)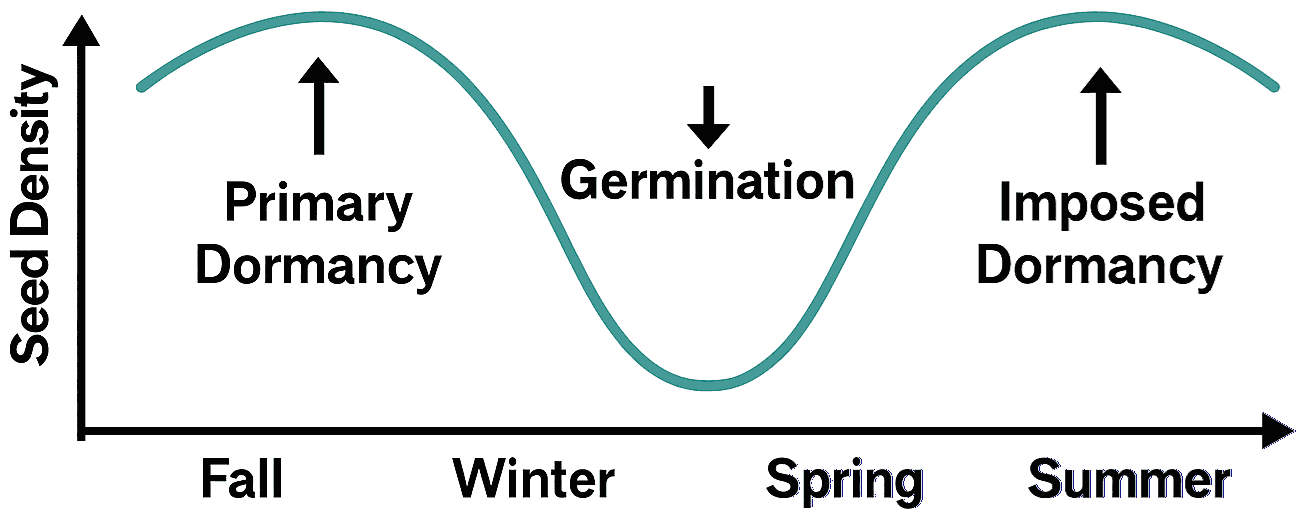 Fig. Seasonal fluctuations in a seebank in temperate regions
Fig. Seasonal fluctuations in a seebank in temperate regionsFall: Seed input increases density; primary dormancy begins Winter: Dormancy maintained; density remains stable Spring: Germination reduces seed density Summer: Imposed dormancy may occur; density slowly recovers Arrows indicate transitions between dormancy states and germination |
Seeder vs sprouterObligate seeder: Regeneration of the rare Morro manzanita (Arctostaphylos morroensis) is dependent on fire. This plant is an obligate seeder; that is, individuals do not resprout after fire, and the species maintains itself solely by reproducing from seed.⇔ Obligate sprouter |
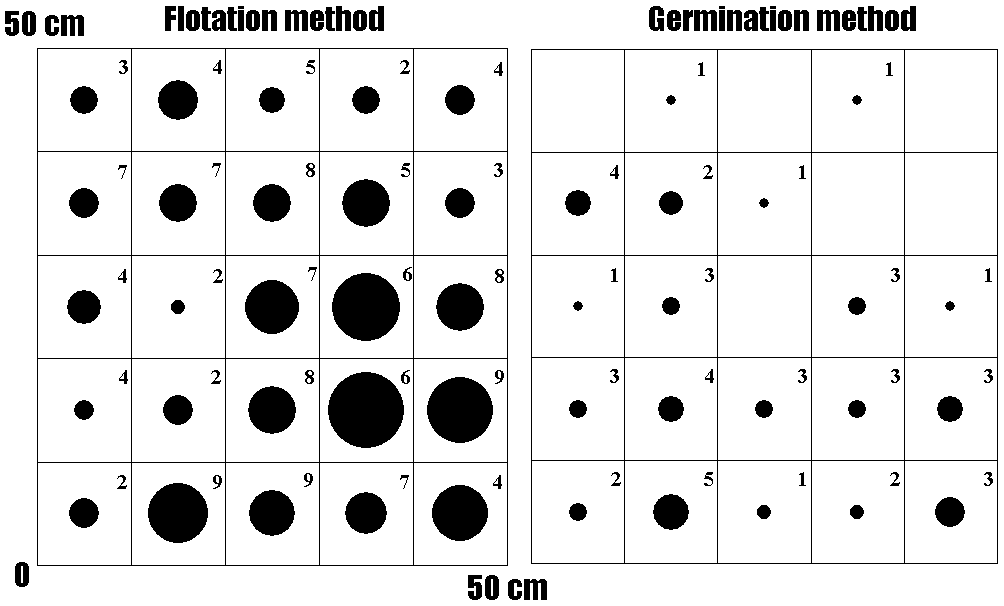 Fig. 1. Spatial distribution of total number of seeds in a 50 × 50 × 10 cm quadrat of which density was the highest in the four quadrats surveyed. The sizes of circles indicate that number of seeds (flotation method) or seedlings (germination method) in each 10 x 10 cm subquadrat. The maximum circle indicates 52 seeds. Numeral within each box represents number of species. (Tsuyuzaki & Goto 2001) • Carex oxyandra • Hypericum erectum • Luzula capitata • Rumex obtusifolius |
 1 1
 2 2
[1] seeds of dominant species (Rumex obtusifolius, and others) in the former topsoil 20 years after the 1977-78 eruptions on Mount Usu (Goto M). The seeds were extracted by a centrifuged floatation method (Tsuyuzaki 1994). [2] how to dig! (April 29 1988) |
Table 1. Types of seedbank estimation (After Tsuyuzaki 1990)
(Feast & Robert 1973) Germination testfor the estimation of the density of buried seedsWeigh soil and place into earthenware dishes (10 cm in diameter)↓ soil thickness = 0.5 cm (ca. 40 cm³) and 1.0 cm (ca. 80 cm³) ↓ three replications = 40 × 3 + 80 × 3 = 360 cm³ Place into an incubator ↓ Add water as necessary ↓ Stirs sample soils every 2 months (not necessarily) Count number of seeds germinated Improved germination testWashing soil samples by coarse and fine sieves
Coarse = 4.00 mm mesh width [Sample soils = clay, peaty, and sandy soils] | Spreading in a 3-5 mm thick layer on sterilized potting compost
|
Concentrating samples can lead to seed losses in soil bank estimationsConcentration, CTraba et al. (1998) = 0.100 mm-diameter sterile nylon mesh [ter Heerdt et al. (1996) = 0.212 mm in mesh width] vs Non-concentration, NCSeedling emergence (/432 cm³)C__199.7 ± 79NC_161.5 ± 11.8 P < 0.001 (paired t-test, n = 10) No significant difference in the number of species per sample (P = 0.153, n = 10) Seven species (Crassula tillaea, Hernaria hirsuta, Sagina apetala, Teesdalia coronopifolia, Trifolium arvense, Tuberaria gutata and Vulpia spp.)
NC > C (Wilcoxon range test, P < 0.05) Discriminant criteria on seed survival (種子生死判別)TTC staining on seed embryo for viability checkProcedure: 0.6% TTC solution (pH 6-7 adjusted by 20 mM K2HPO4 or K2HPO4) → Cut seed vertically → Place them for 24 hr in the dark → Inspect the embryo under a binocular stereomicroscopePreparation of artificial buried-seed populations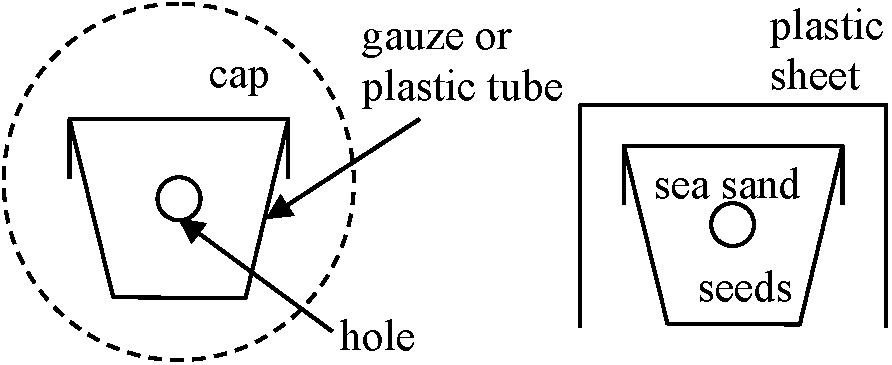 Seeds ↓ mixed with sea sand (not necessarily) ↓ Packed into a holed plastic tube Seeds burial into soil at any soil depths Small plastic capsules, each of which contains 50 seeds and sea sand, are buried into soil. Four pin holes were drilled through each capsule so that the contents of air and humidity might equilibrate with those outside. |
Seed extraction from soils by a flotation method (Tsuyuzaki 1994)Abstract. In this study a flotation method is described which enables the rapid extraction of seeds from soil. Essentially, soil is mixed with 5.5 M K2CO3, and thoroughly dispersed by mechanical stirring for 3-6 min. The dispersed soil samples were delivered to plastic tubes, then centrifuged at more than 4,000 g for about 5 min. Buried seeds and lighter organic materials floated in the supernatant and could be removed by filtration, washed and subsequently identified. This method performed reliably using soil samples artificially burdened with white clover and a range of cultivated grass species. In addition, a wide range of weed seeds which differed in their size and densities were also effectively recovered from a range of soil types loam to gravel.PrincipleSeeds are floated by a high-concentration solution. We use 50% K2CO3 solution with cnetrifugation described below. This method is improved gradually.Flotation method with centrifugationSoil samples 1
↓ - add 50% K2CO3 (specific gravity = 1.54)
↓ - filtration using an aspirator ³ [Remarks]
|
PreparationFor flotationPortable centrifugatorCentrifugation tubes (> 200-ml is nice) Floatation solution (see below) Distilled water Beakers (size is dependent on the volume of solution) Bottle with nozzle (to wash organic debris) Pipette Balance (for centrifugation) Miracloth Tweezers Stirrer (shaft is better) Aspirator Buchner funnel Suction bottle Rag (floorcloth), and/or Kim-towel Rubber gloves (if your skin is sensitive) Flotation solution50% K2CO3 (Tsuyuzaki 1993)Add 770 g K2CO3 by bits to 1 l H2O in a beaker with stirring by using a mechanical stirrer → Appplication: (Tsuyuzaki 1989, Tsuyuzaki 2010, Ishikawa-Goto & Tsuyuzaki 2004) Calgon mixture (Malone 1967) 10 g sodium hexametaphosphate (calgon, ヘキサメタリン酸ナトリウム), (NaPO3)nP2O5 5 g sodium bicarbonate (baking soda, 炭酸水素ナトリウム) NaHCO3 25 g magnesium sulphate (epsom salts, 瀉痢塩) MgSO4 200 mg tap water For sortingBinocular stereomicroscopeTweezzers Needle Petri dishes Of course, notebook and writing tools |
|
(Yamasaki et al. 2020) Genebank ジーンバンク (農研機構)_______Estimated seed longevity (year)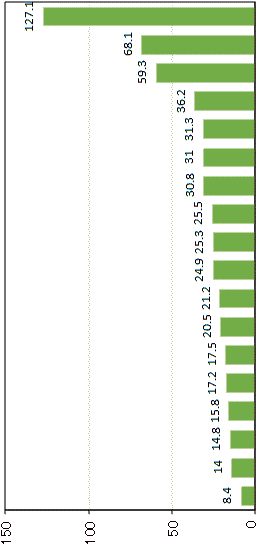 Cucumis sativus
Cucumis sativusFagopyrum esculentum Cucumis melo Solanum melongena Solanum lycopersicum Hordeum vulgare var. hexastichon Glycine max ssp. soja Sorghum bicolor Oryza sativa (southern Asia) Oryza sativa (southeastern Asia) Zea mays ssp. mays Triticum aestivum Setaria italica Oryza sativa Glycine max Oryza sativa (eastern Asia) Vigna angularis var. angularis Megathyrsus maximus |
Dr. Beal's experimentsTable. Results of Dr. Beal's experiments (%)
Year 50 80 100 |