(Upload on June 21 2025) [ 日本語 | English ]
Mount Usu / Sarobetsu post-mined peatland
From left: Crater basin in 1986 and 2006. Cottongrass / Daylily
HOME > Lecture catalog / Research summary > Glossary > Biology > Microbiology
|
microbe < 100 μm = bacteria + archaea (+ fungi + eukaroyotes)
bacteria = procaryotae (原核生物) + monera (= mychota, anucleobionta) Endophyte (エンドファイト): endo = within + phyte = plant (L.)bacteria and fungi living symbiotically within a plant for at least part of their life cycles
Endosphere: all the endophytes of a plant Gram stain (グラム染色)= Gram staining and Gram's methodClassified into gram-positive and gram-negative bacteria by staining ⇒ the chemical and physical (morphological) properties of cell envelopes and walls Gram-positiveGram-negative + Gram-variable and Gram-indeterminate bacteria Morphology (形態)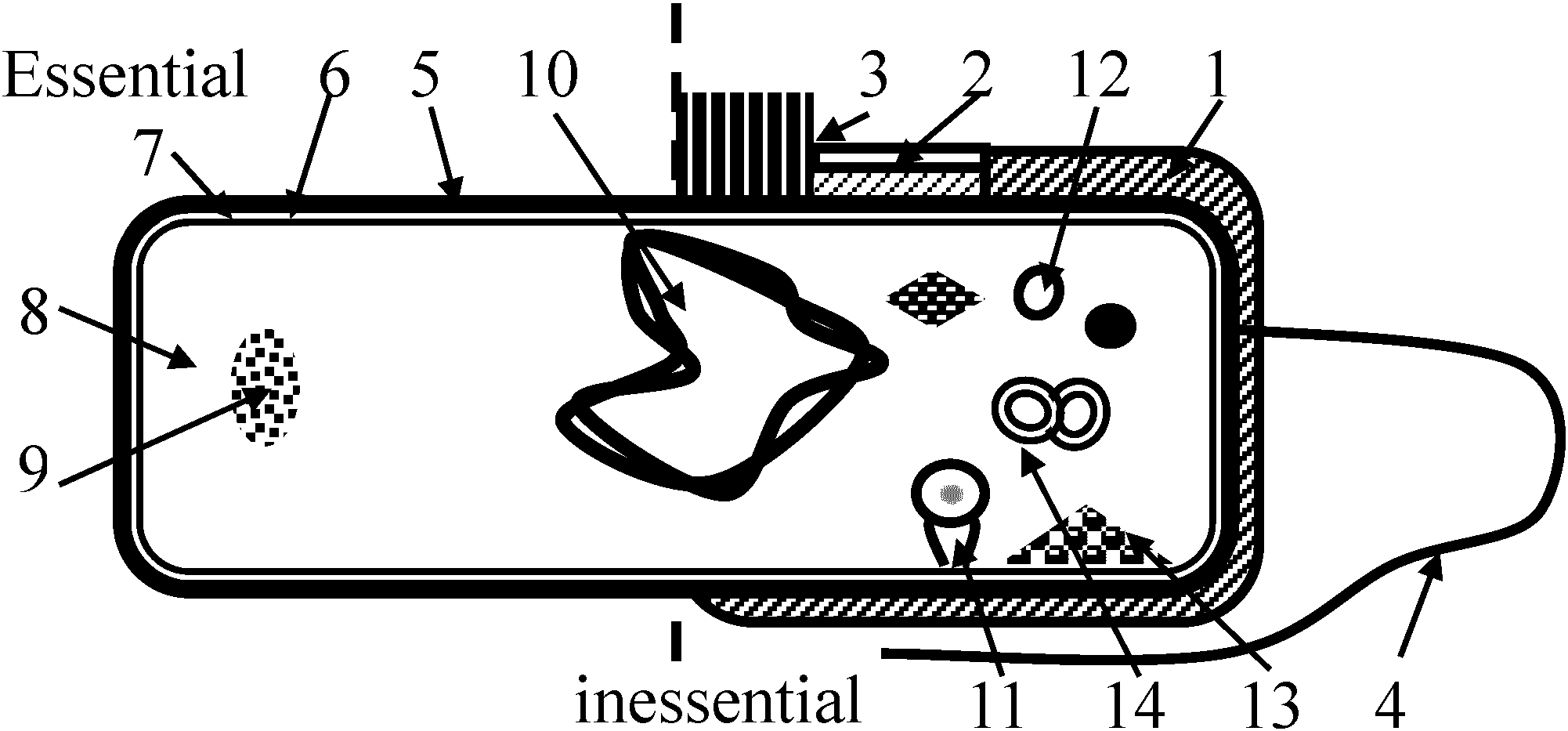 Fig. 1: capsule, slime layer, 2: microcapsule, 3: pili, fimbriae, 4: flagellum, 5: cell wall, 6: cytoplasmic membrane (cell membrane, plasma membrane), 7: periplasmic space, 8: cytoplasm, 9: ribosome, 10: bacterial chromosome (double-strand attached with PM), 11: mesosome, 12: vacuole, 13: granules, 14: plasmid 1. Capsules and slime layer2. Flagellum (pl. -a)Polarity (極性): single polar, monotrichous, monopolar, dipolar, several polar, polytrichous polar, multitrichous, subpolar, lateral, peritrichousTerminology: acronematic (adj.) referring to a flagellum that is smooth and terminated with a fine fibril, thus tapered at its distal end 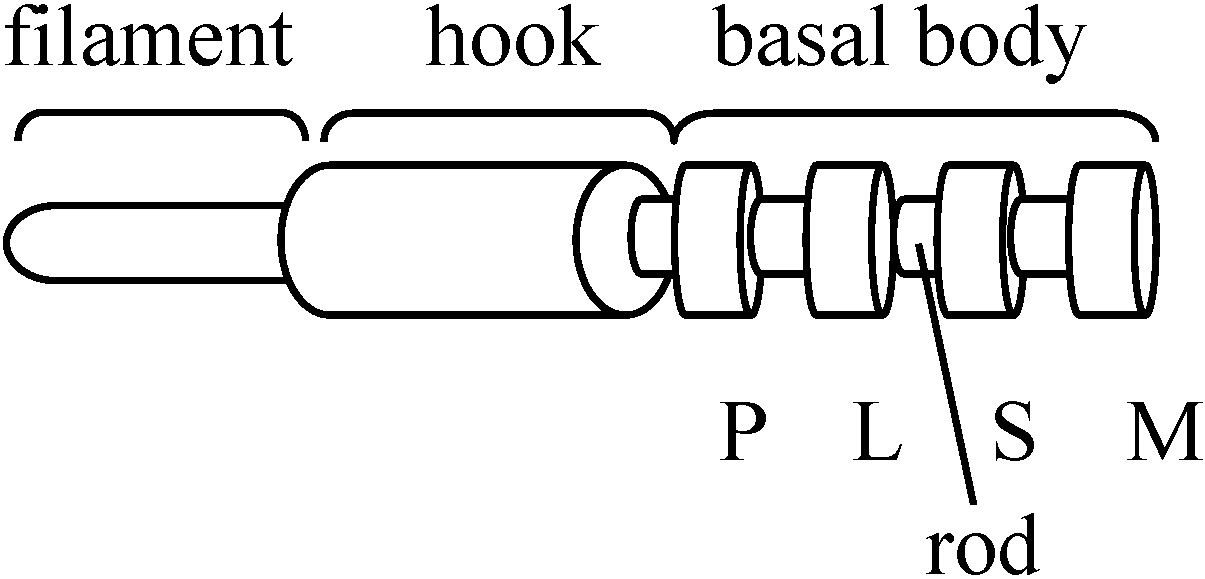 Structure: three components
Structure: three componentsBasal body P ring, L ring, S ring, M ring + rod Hook (フック) = function as universal joint (自由継手)
the diameter of hook is larger than that of filament flagellin (フラジェリン) = subunits, consisting of proteins, that form filaments - recovered by centrifugation
Species ______________ Subunit MW
B. subtilis flagellin: lacking four amino acids, Try, Cys, Asn and Glu, and consisting of 16 kinds of amino acids (Rhizobium meliloti, the same with B. subtilis)
C. aecsentus 25 kD, 27.5 kD strain → flagellin → [differentiation] → one phase / two phase
Ex. put two types of flagella into a test tube → [plate culture] → obtaining two types of colonies 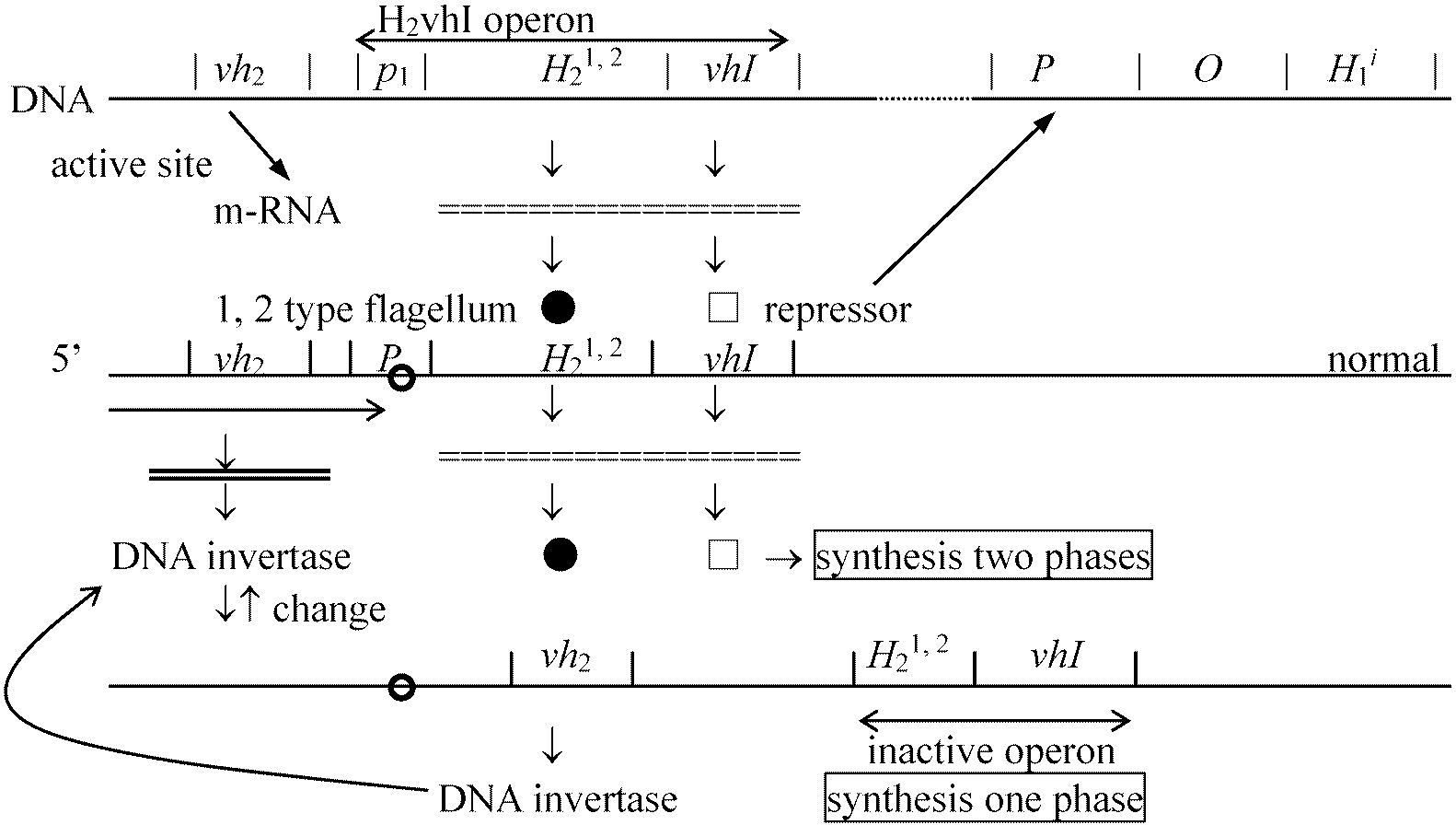 3. Pili (繊毛)a) bacterial fimbria or common piliTable. Types and characteristics of pili. *: after tannic-acid treatment
Size
Red
blood
Response
Note that the responses differ when the red blood originatig from cow and chiken |
affected by differences in the sequence of amino acids pili → [heat treatment] → subunits = subunit of finbice???b) sex pili (or fimbriae): firstly discovered from E. coli male straing - female strain 4. nucleus (in prokaryotes) (核)5. cytoplasmic granules (細胞内顆粒)6. cytoplasm (サイトプラスム)7. mesosome (メソゾーム)= chondrioidInvaginations in the plasma membrane of bacteria - concentrical multi-layred membrane 1-3 mesosomes/cell - changing with culture condition (< 40/cell) 1953 Chapman J & Hillier J: reported peripheral bodiesfrom gram-positive bacteria 1959 Fitz-James JD: termed mesosomeAfter discovering mesosome, the function was discussed
H1: analog of mitochondria in eukaryotes P1: most gram-negative bacteria do not form mesosome. Mesosomes are reported from only three taxa (Caulobacter aresceutus, Flexibacter columnaris, Chromobacterium violaceum) 1971 Nanninga: experiment that demonstrates mesosomes are artifact
Fixed chemically__Frozen______Fixed then frozen 8. (cell) envelopes (細胞外皮)= cell wall + periplasm (including periplasmic space) + peptidoglycan + cell membrane OCMBasic structure of cell envelopesA) Membrane modelB) Cytoplasmic membrane C) Outer membrane Molecular shield (分子盾)
cf. mitochondria → nucleus →chlorophyll (N-end) Cys1-Ser2-Ser3- … -Lys58 (C-end)
the cells treataed by toluene (C6H5CH3) that uncouples lipoprotein → mRNA (LP) is synthesized in vitro Extra-peptide = leader peptide or signal peptide D) Periplasmic space or periplasm E) Adhesion zone 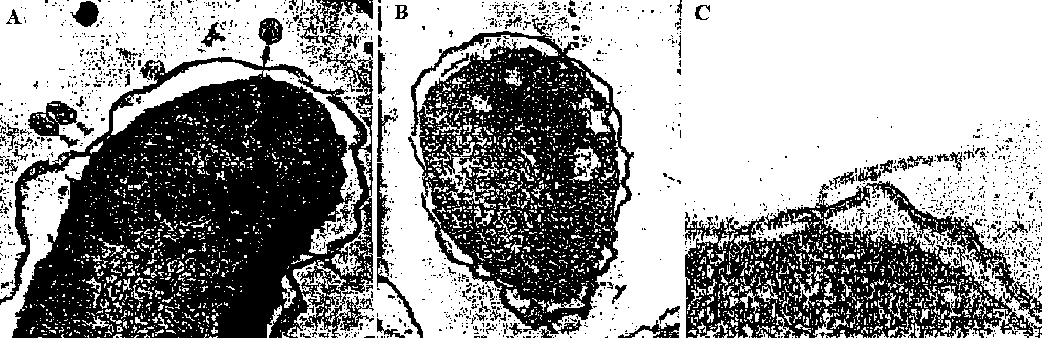 Fig. A: Bacteriophage T2 adsorbed to a plasmolized E. coli B cell. The virus part with their tail tips (= cores) to the adhesion sites. B: Ultrathin section through E. coli HfrH, revealing the F-pilus insertion at the membrane adhesion zone. The pilus is labeled with bacteriophage MS2. C: Ultrathin section of E. coli showing a flagellum insertion area. Note the apparent organization of the inner membrane at the insertion point. L-form bacteria (L型菌)= L-phase bacteria, L-phase variants and cell wall-deficient (CWD) bacteria1935 Klieneberger-Nobel E (1892-1985) discovered strains of bacteria that lack cell walls |
[ nomenclature ]
s.s. prokaryotes = archaea (archaebacteria) + eubacteria (bacteria)
s.l. prokaryotes (s.s.) + viroids + viruses
Morphology (at microscope level)
coccus (球菌), bacillus (棹菌), coccobacillus (球桿菌), vibrio (コンマ菌), spirillum (螺旋菌), spirochaete (スピロヘータ), streptomyces (ストレプトミセス), crenothrix (クレノスリクス) |
IMViC test (インビック試験)= Indole test + Methyl red test + Voges-Proskauer test + Citrate test1. Indole reaction Trypthane → Tryptophanase → Indole → p-Dimethylaminobenzaldehyde → Red Indole Medium: 1-2% Peptone, 0.5% NaCl (pH = 7.2)↓ Autoclave, Inoculation at 24-37°C, Incubation for 2-7 days ↓ + Kamacs indole regent Kamacs indole regent: amylalhol (cis-amylalchol) = 150 ml, D-dimethyaminobenyaldehyde = 10 g, conc = 50 ml 2. methyl red reaction
Medium: Glucose K2HPO4 Peptone Distilled water
→ autoclave at 115°C for 15 min.
Glucose |
Mineral salts (無機塩類)preserve the structure of bacteria
|
1970s' Woose Carl (1928-2012): proposed classification based on rRNA
Kingdom Prokaryote 原核生物界Domain Bacteria 細菌 (Eubacteria 真正細菌)coccus - bacillusPhylum AquificaeAquificaceaeDesulfurobacteriaceae Hydrogenothermaceae Phylum ThermotogaeThermotogaceae: Thermotoga maritima, optimal temeprature for the reproduction = 80°C (reproduced at 90°C)Phylum ThermodesulfobacteriaThermodesulfobacteriaceaePhylum Deinococcus-ThermusOrder DeinococcalesDeinococcaceae: Deinococcus, DeinobacteriumTrueperaceae: Truepera Order ThermalesThermaceae: Thermus, Marinithermus, Meiothermus, Oceanithermus, VulcanithermusPhylum ChrysiogenetesChrysiogenes arsenatis, Desulfurispirillum alkaliphilumPhylum Chloroflexi (緑色滑走細菌)Class ChloroflexiOrder ChloroflexalesChloroflexaceae (緑色糸状細菌): Chloroflexus, Chloronema, Heliothrix - photosynthesisOscillochloridaceae: Oscillochloris Order HerpetosiphonalesHerpetosihpnaceae: HerpetosihponPhylum ThermomicrobiaPhylum NitrospiraPhylum DeferribacteresPhylum CyanobacteriaPhylum Chlorobi (クロロビウム)Class ChlorobiaOrder ChlorobialesChlorobiaceaeChlorobium (green sulfur bacteria, 緑色硫黄細菌), Ancalochloris, Chlorobaculum, Chloroperpeton, Pelodictyon, Prosthecochloris Phylum Proteobacteria (プロテオバクテリア)Class α-proteobacteriaOrder RhodospirillalesRhodospirillaceae (formerly Athiorhodaceae) 紅色非(無)硫黄細菌Azospirillum, Rhodospirillum, Rhodopseudomonas, Rhodomicrobium Acetobactaceae Acetobacter (A. aceti 酢酸菌) Order Rickettsiales (リケッチア)Rickettsiaceae: RickettsiaEhrlichiaceae Holosporaceae Anaplasmataceae Philip 1957 (アナプラズマ) Anaplasma Theiler 1910 Ehrlichia Neorickettsia Wolbachia Hertig 1936: symbiont with invertebrates (arthropods) and filariae sex-ratio distorters (male-killing, parthenogenesis, feminization, etc.) Order RhodobacteralesOrder SphingomonadalesSphingomonadaceae: SphingomonasOrder CaulobacterialesCaulobacteraceae Henrici et Johnson 1935Asticcacaulis, Brevundimonas, Caulobacter, Phenylobacterium Order Rhizobiales (リゾビウム)(Hyphomicrobiales, discarded)Rhizobiaceae Rhizobium Frank 1989: nodule, nitrate bacteria rod, gram-negative, aerobic, not sporulated Agrobacterium, Carbophilus, Chelatobacter, Ensifer, SinorhizobiumBartonellaceae Brucellaceae Phyllobacteriaceae Methylocystaceae Beijerinckiaceae: Beijerinckia, Camelimonas, Chelatococcus, Methylocapsa, Methylocella, Methyloferula, Methylorosula, Methylovirgula, Pseudochelatococcus Nitrobacteraceae, corrig. Buchanan 1917 (incl. Bradyrhizobiaceae) Variibacter, Pseudorhodoplanes, Pseudolabrys, Afipia, Rhodopseudomonas, Bradyrhizobium Nitrobacter: NO2 + 1/2O2 → NO3 (oxidation of NO2 - N cycle) HyphomicrobicaceaeMethylobacteriaceae Rhodobiaceae Class β-proteobacteria (ベータプロテオバクテリア)gram-negativeOrder BurkholderialesBurkholderiaceae: Burkholderia, Cupriavidus, Lautropia, Pandoraea, ThermothrixRalstoniaceae: Ralstonia Oxalobacteraceae: Janthinobacterium De Ley et al. 1978, emend. Lincoln et al. 1999: soil bacteria (J. agaricidamnosum Lincoln et al. 1999, J. lividum De Ley et al. 1978) Alcaligenaceae De Ley et al. 1986: found in water, soil and animals Alcaligenes Castellani et Chalmers 1919 Achromobacter Yabuuchi et Yano 1981, aerobic in water: A. fisheri Bordetella Moreno-López 1952: B. pertussis Comamonadaceae: Comamonas, Acidovorax, Aquabacterium, Brachymonas, Delftia, Hydrogenophaga, Ideonella, Leptothrix, Polaromonas, Rhodoferax, Roseateles, Rubrivivax, Sphaerotilus (S. nataus), Tepidimonas, Thiomonas, Variovorax Order HydrogenophilalesOrder MethylophialesOrder NeisserialesNeisseriaceae: Neisseria (N. grorrhoeae), Alysiella, Aquaspirillum, Catenococcus, Chromobacterium, Eikenella, Formivibrio, Iodobacter, Kingella, Microvirgula, Prolinobarus, Simonsiella, Vitreoscilla, VogesellaOrder NitrosomonadalesNitrosomonadaceae: Nitrosolobus, NitrosospiraNitrosomonas: NH3 + 1·1/2O2 → NO2 + H2O + 2H (oxidation of NH4) Methylophilaceae: Methylophilus, Methylobacillus, MethylovorusSpirillaceae: Spirillum Thiobacillaceae: Thiobacillus, Annwoodia, Sulfuritortus Gallionellaceae: Gallionolla (G. ferginea), Ferriphaselus, Sulfuriferula, Sulfurirhabdus, Sulfuricella Sterolibacteraceae: Sterolibacterium, Sulfurisoma, Denitratisoma, Sulfuritalea, Georgfuchsia, Sulfurisoma, Methyloversatilis Order RhodocyclalesRhodocyclaceaeAzonexaceae Zoogloeaceae Class γ-proteobacteriaOrder ChromatialesChromatiaceae: Chromatium, Allochoromatium, Amoebobacter, Halochromatium, Halothiobacillus, Isochromatium, Lamprobacter, Lamprocystis, Marichromatium, Nitrosococcus, Pfennigia, Rhabdochromatium, Thermochromatium, Thioalkalicoccus, Thiocapsa, Thiococcus, Thiocystis, Thiodictyon, Thiohalocapsa, Thiolamprovum, Thiopedia, Thiorhodococcus, Thiorhodovibrio, ThiospirillumEctothiorhodospiraceae: Ecthiorhodospira, Arhodomonas, Halohodospira, Nitrococcus, Thiorhodospira Order ActithiobacillalesOrder XanthomonadalesXanthomonadaceae: Xanthomonas, StenotrophomonasOrder CardiobacterialesOrder ThiotrichalesThiotrichaceae: Thiothrix, Achromatium, Beggiatoa, Leucothrix, Macromonas, Thiobacterium, Thiomargarita, Thioploca, ThiospiraPiscirickettsiaceae Francisellaceae Order LegionellalesLegionellaceae: Legionella ALCoxiellaceae: Coxiella, Rickettsiella Order MethylococcalesOrder OceanspirillalesOrder Pseudomonadales (シュードモナス)Pseudomonas Migula 1894 (syn. Chlorobacterium), Azomonas, Azotobacter (A. chroocaccum), Cellvibrio, Chryseomonas, Flavimonas, Lampropedia, Mesophilobacter, Morococcus, Oligella, Rhizobacter, Rugamonas, Serpens, Thermoleophilum, XylophilusMoraxellaceae: Moraxella, Acinetobacter Moraxellaceae Order AlteromonadalesAlteromonadaceae: Alteromonas, ShewanellaOrder VibrionalesVibrionaceae: Vibrio (Ex. Cholera group) (V. möller, V. comma, V. parahaemelyticusOrder AeromonadalesAeromonadaceae: AeromonasOrder EnterobacterialesEnterobacteriaceae (腸内細菌科)Order PasteurellalesPasteurellaceae: Pasteurella, Actinobacillus, HaemophilusClass δ-proteobacteria, nom. nud. (δ-プロテオバクテリア)Syntrophorhabdaceae, order not decidedOrder BdellovibrionalesBacteriovoracaceaeBdellovibrionaceae Bdellovibrio Stolp and Starr Order DesulfarculalesDesulfarculaceaeOrder DesulfobacteralesDesulfobacteraceaeDesulfobulbaceae Nitrospinaceae Order DesulfovibrionalesDesulfohalobiaceaeDesulfomicrobiaceae Desulfonatronaceae Desulfovibrionaceae Desulfovibrio Kluyver et van Niel 1936: gram-netative, nitrate-reducing Order DesulfurellalesDesulfurellaceaeOrder Desulfuromonadalesanaerobic respiration uisng various compounds as electron acceptors, including sulfur, Mn(IV), Fe(III), nitrate, Co(III), Tc(VII), U(VI) and trichloroacetic acidDesulfuromonadaceae, corrig. Kuever et al. 2006 Geobacteraceae Holmes et al. 2004 Geoalkalibacter Zavarzina et al. 2007 Geobacter Lovley et al. 1995 related to the diffusion of uranium - bioremediation Geopsychrobacter Holmes et al. 2005Geothermobacter Kashefi et al. 2005 Pelobacteraceae fermentative metabolism Order Myxobacteriales (slime bacteria, 粘液細菌)Suborder CystobacterineaeCystobacteraceaeMyxococcaceae Myxococcus xanthus Suborder SorangiineaeMinicystis rosea Garcia et al. 2014, family not decided: the lrageest genome size (16,040,666 bp) in prokaryotes (by 2018)Polyangiaceae Phaselicystidaceae Sandaracinaceae Suborder NannocystineaeNannocystaceaeKofleriaceae Haliangiaceae Order SyntrophobacteralesSyntrophaceaeSyntrophobacteraceae Class ζ-proteobacteriaPhylum Firmicutes (グラム陽性細菌門)Class Bacilli (バシラス)Order Bacillales (バシラス)Bacillaceaegram-positive, heterotrophic, rod-shaped bacteria that may produce endospores Bacillus Cohn 1872 Order Lactobacillales (ラクトバシラス)Aerococcaceae (アエロコックス)gram-positive Abiotrophia, Aerococcus (A. viridans: widespread - causative agent of gaffkaemia, a disease of lobsters), Dolosicoccus, Eremococcus, Facklamia, Globicatella, Ignavigranum Carnobacteriaceae (カルノバクテリウム) Granulicatella, Aerococcus, Alloiococcus, Abiotrophia, etc. Enterococcaceae (エンテロコッカス) Enterococcus, Melissococcus, Pilibacter, Tetragenococcus, Vagococcus Lactobacillaceae Pediococcus Leuconostocaceae (ロイコノストック), formerly grouped into Lactobacillaceae Leuconostoc: L. mesenteroides, wine fermentation Fructobacillus, Oenococcus, Weissella, etc. Streptococcaceae (連鎖球菌) formerly Enterococcus (腸球菌) was categorized into this family (now Enterococcaceae) Streptococcus Rosenbach 1884: S. pneumoniae (肺炎球菌), S. viridans (緑色連鎖球菌) Floricoccus Lactococcus Schleifer et al. 1986: lactic acid bacteria Lactovum Okadaella Streptococcus Genera incertae sedis: Aerosphaera, Carnococcus, Chungangia Class Clostridia (クロストリジウム)Order Clostridiales (クロストリジウム)gut bacteria (gb)Caldicoprobacteraceae Caldicoprobacter: C. oshimai, C. algeriensis, C. guelmensis, C. faecale (syn. Acetomicrobium faecale) |
Christensenellaceae (human gb) Christensenella minuta Clostridiaceae (クロストリジウム) Clostridium Prazmowski 1880: C. tetani (Fl&umul;gge 1886) Bergey et al. 1923 (破傷風菌), C. botulinum, C. pasteurianum Winogradsky 1895 (syn. C. pastorianum): fixing free nitrogen from the air, C. perfringens (Veillon & Zuber 1898) Hauduroy et al. 1937 (ウェルシュ菌) (facultative human gb) Dehalobacteriaceae Dehalobacterium (gb) Defluviitaleaceae Eubacteriaceae Ludwig et al. 2010 (ユーバクテリウム) Acetobacterium Balch et al. Eubacterium Prévot 1938: hard cell wall Graciibacteraceae Heliobacteriaceae Lachnospiraceae (ラクノスピラ) (gb) Anaerostipes (human gb), Blautia: B. coccoides, Butyrivibrio, Coprococcus, Dorea, Lachnospira Oscillospiraceae Peptococcaceae (ペプトコッカス) Desulfosporosinus Stackebrandt et al. 1997 - soil and groundwater Peptostreptococcaceae Garrity et al. 2001 (gb) Acetoanaerobium, Clostridioides, Filifactor, Peptostreptococcus, Tepidibacter, etc. Ruminococcaceae (ルミノコッカス) Ethanoligenens, Faecalibacterium (F. prausnitzii, gb), Ruminococcus Sijpesteijn 1948, Oscillospira Sulfobacillaceae Sulfobacillus Syntrophomonadaceae Zhao et al. emend, Jumas-Bilak et al. 2009 using carboxylic acids as a source of energy Syntrophomonas McInerney et al. 1982 Veillonellaceae Rogosa 1971 (= formerly Acidaminococcaceae) obligate anaerobes in rivers, lakes, and the intestines of vertebrates Veillonella Prévot 1933 (12 spp.), Megasphaera (5), Dialister (ex Bergey et al. 1923) Moore and Moore 1994 (5), Allisonella (A. histaminiformans): ruminal, Anaeroglobus Carlier et al. 2002 (A. geminatus), Negativicoccus Marchandin et al. 2010 (N. succinicivorans) Order HalanaerobialesHalanaerobiaceaeHalobacteroidaceae Order NatranaerobialesNatranaerobiaceaeOrder ThermoanaerobacterialesThermoanaerobacteraceaePolyphyletic Thermodesulfobiaceae Mori et al. 2004 Class Erysipelotrichi (エリュシペロトリクス)Order ErysipelotrichalesClass Thermolithobacteria Sokolova et al. 2007Order ThermolithobacteralesThermolithobacteraceae (テルモリトバクテル)Class Limnochordia Watanabe et al. 2015 (リムノコルダ)Class Tissierellia ティッセレッラ(Acidaminococcaceae)Phylum Tenericutes or Firmicutes (テネリクテス)Class Mollicutes (モリクテス)lacking cell wall - smallparasites to plants and animals not stained by Gram staining Order Mycoplasmatales (マイコプラズマ)cell wall-less (and virus-less)Mycoplasmataceae Freundt 1955 Mycoplasma J Nowak 1929: over 100 species Ureaplasma: 8 species so far, urease positive Order Entomoplasmatales (エントモプラズマ)Entomoplasmataceae: Entomoplasma, Mesoplasma Spiroplasmataceae (スピロプラズマ): small without cell wall, insect symbiontSpiroplasma: S. citri - citrus stubborn disease, S. kunkelii - corn stunt disease Order Acholeplasmatales (アコレプラズマ)AcholeplasmataceaeCa. Phytoplasma Firrao et al. (ファイトプラスマ): cell wall-less, mycoplasma-like organism (MLO) - plant deseases Order Anaeroplasmatales (アナエロプラズマ)AnaeroplasmataceaeOrder Haloplasmatales (ハロプラズマ)Haloplasmataceae: Haloplasma (H. contractile = halophilic, cell wall-less)Phylum Actinobacteria (アクチノバクテリア)Gram-positive, including high DNA GC contentClass ActinobacteriaSubclass Actinobacteridaeactinorrhizal: adj. 放線菌の Order Actinomycetales (放線菌)Formerly characterized by the development of myceliaMycelium = vegetative (or substrate) mycelium + aerial mycelium Suborder ActinomycineaeActinomycetaceae: Actinomyces, Actinobaculum, Arcanobacterium, MabiluncusSuborder CorynebacterineaeCorynebacteriaceae: CorynebacteriumDietziaceae: Dietzia Gordoniaceae: Gordonia, Skermania Mycobacteriaceae (マイコバクテリウム) Fast-growing mycobacteria
Mycobacteroides
Mycolicibacter Tsukamurellaceae: Tsukamurella Williamsiaceae: Williamsia Subclass RubrobacteridaeOrder RubrobacterialesSuborder RubrobacterineaeRubrobacteriaceae: RubrobacterSubclass CoriobacteridaeOrder CoriobacterialesSuborder CoriobacterineaeCoriobacteriaceae: Coriobacterium, Atopobium, Collinsella, Cryptobacterium, Denitrobacterium, Eggerthella, SlackiaSubclass SphaerobacteridaeOrder SphaerobacteralesSuborder SphaerobacterineaeSphaerobacteraceae: SphaerobacterSuborder FrankineaeFrankiaceae (フランキア) ☛ nitrogen fixationFrankia Brunchorst F. alni (Woronin) Von Tubeuf: host = alder, no flagella and mobility Phylogenetic analyses → divided into four genera = Frankia +
Protofrankia + Parafrankia + Pseudofrankia Microsphaeraceae: Microsphaera Sporichthyaceae: Sporichthya Acidothermaceae: Acidothermus Kineosporiaceae: Kineosporia, Cryptosporangium, Kineococcus Suborder MicrococcineaeParaphyleticMicrococcaceae: Micrococcus, Arthrobacter, Kocuria, Nesterenkonia, Renibacterium (moved), Rothia, Stomatococcus Bogoriellaceae: Bogoriella (moved) Rarobacteraceae: Rarobacter (moved) Sanguibacteraceae: Sanguibacter Brevibacteriaceae: Brevibacterium Cellulomonadaceae: Cellumonas, Oerskovia Dermabacteraceae: Dermabacter, Brachybacterium Dermatophilaceae: Dermatophilus Dermacoccaceae: Dermacoccus (moved), Demetria (moved), Kytococcus (moved) Intrasporangiaceae: Intrasporangium, Janibacter, Ornithinicoccus, Ornithinimicrobium, Nostocoidia, Terrabacter, Terracoccus, Tetrasphaera Jonesiaceae: Jonesia Microbacteriaceae: Microbacterium, Agrococcus, Agromyces, Aureobacterium, Clavibacter, Cryobacterium, Curtobacterium, Frigoribacterium, Leifsonia, Leucobacter [moved], Rathayibacter, Subtercola Beutenbergiaceae: Beutenbergia Promicromonosporaceae: Promicromonospora Suborder MicromonosporineaeMicromonosporaceae: Micromonospora, Actinoplanes, Catellatospora, Catenuloplanes, Couchioplanes, Dactylosporangium, Pilmelia, Spirilliplanes, VerrucosisporaSuborder PropionibacterineaePropinibacteriaceae: Propinibacterium, Luteococcus, Microlunatus, Propioniferax, TessaracoccusNocardioidaceae: Nocardioides, Aeromicrobium, Friedmanniella, Hongia, Kribbella, Micropruina, Marmoricola Suborder PseudonocardineaePseudonocardiaceae: Pseudonocardia, Actinoalloteichus, Actinopolyspora, Amycolatopsis, Kibdelosporangium, Kutzneria, Prauserella, Saccharopolyspora, Streptolloteichus, Thermobispora, ThermocrispumActinosynnemataceae: Actinosynnema, Actinokineospora, Lentzea, Saccharothrix Suborder StreptomycinaeStreptomycetaceae: Streptomyces (S. griseus), Kitasatospora, StreptoverticilliumSuborder StreptosporangineaeStreptosporangiaceae: Streptosporangium, Acrocarpospora, Herbidospora, Microbispora, Microtetraspora, Nonomuraea, Planobispora, Planomonospora, Planopolyspora, PlanotetrasporaNocardiopsaceae: Nocardiopsis, Thermobifida Thermomonosporaceae: Thermomonospora, Actinomadura, Spirillospora Order Bifidobacteriales (ビフィドバクテリウム)gram-positive, obligately anaerobicBifidobacteriaceae: Aeriscardovia, Alloscardovia, Bifidobacterium Orla-Jensen 1924 (ビフィズス菌), Bombiscardovia, Galliscardovia, Gardnerella, Neoscardovia, Parascardovia, Pseudoscardovia, Scardovia A few unknown affilitation: Actinobispora, Actinocorallia, Excellospora, Pelczaria, Turicella Phylum Planctomycetes (プランクトミケス)Class ChlamydiaeOrder ChlamydialesChlamydiaceae: ChlamydiaParachlamydiaceae Simkaniaceae Waddliaceae Phylum SpirochaetesClass SpirochaetesOrder Spirochaetales (螺旋菌/スピロヘータ)Spirochaetaceae: Spirochaeta, Borrelia (B. recurrentis), Brevinema, Clevvelandina, Cristispira, Diplocalyx, Hollandina, Pillotina, Treponema (T. pallida)Serpulinaceae: Serpulina, Brachyspira Leptospiraceae: Leptonema, Leptospira (L. icterohaemorrhagiae) Phylum Fibrobacteres Garrity et Holt 2012 (フィブロバクテル)gram-negativeClass ChitinispirilliaClass ChitinivibrioniaClass FibrobacteriaPhylum Acidobacteria (アシドバクテリウム)Phylum Bacteroidetes (バクテロイデス)= Cytophaga-Flavobacterium-Bacteroidetes (CFB) groupBacteroidetes/Chlorobi group = CFB group + Chlorobi > 350 genera(Class Bacteroidia)Order Bacteroidaleshuman gut microbiotaBacteroidaceae: Bacteroides Rikenellaceae: Rikenella Porphyromonadaceae: Porphyromonas Prevotellaceae: Prevotella Paraprevotellaceae, formerly included in Prevotellaceae Order MarinilabilialesClass Flavobacteriia (フラボバクテリウム)habitat: soil, freshwater, seawaterOrder FlavobacterialesFlavobacteriaceae: Flavobacterium (soil and freshwater/fish - disease), Capnocytophaga, ChryseobacteriumClass Sphingobacteriiaproducing sphingolipids Phylum Sphingobacteria = classes Chlorobea (Chlorobi) + Fibrobacteres + Bacteroidetes + Flavobacteria (proposal - not supported)Order SphingobacterialesSphingobacteriaceae: Sphingobacterium, PedobacterFlexibacteraceae: Flexibacter, Cytophaga Class Cytophagia Nakagawa 2012 emend. Hahnke et al. 2016Phylum Fusobacteria (フソバクテリウム)nearly all species are rod-shapedmost of them are gut bacteria Class FusobacteriiaOrder FusobacterialesFusobacteriaceae: Cetobacterium, Fusobacterium, Ilyobacter, Propionigenium, PsychrilyobacterLeptotrichiaceae: Leptotrichia, Sebaldella, Sneathia, Streptobacillus Phylum Verrucomicrobia (ウェルコミクロビウム)habitats are diverseClass VerrucomicrobiaeClass OpitutaePhylum Dictyoglomi Saiki et al. 1985 (ディクチオグロムス)consisting a single genus that is: Dictyoglomusgram-negative, with a triple-layered wall survived at > 80°C Order DictyoglomalesDictyoglomaceae: D. thermophilum |
Microbial community (微生物群集)Analytical methods16S/18S rRNA gene amplicon sequencing analysis↓ DNA extraction ↓ PCR amplification ↓ DNA sequencing Pre-analysis (remove primers, chimera, etc.) ↓ Sequene clustering at a taxon level by CD-HIT-EST, UCLUST, etc.Taxonomical assignment and analysis Metagenomic sequencing analysis ↓ DNA extraction ↓ DNA sequencing ↓ Metagenomic reads ↓ Assemble: MEGAHIT, MetaPlatanus, IDBA-UD, etc. ↓ Contig sets ↓ Gene finding: MGS, MetaGeneMark ↓ Gene sets ↓ BLASTP → Comparative metagenomics (metadata)
Gene function abundance PipelineQIIME 2: free, open source= Quantitative Insights Into Microbial Ecology Mothur USEARCH |
Microbiota (微生物叢)= formerly microbiol flora, because bacteria were grouped into plantsTable. Major microbiota assemblages found in wetlands.
Quarum sensing (quorum signalling, QS, 集団感知)the ability to detect and respond to cell population density by gene regulationSymbiosomevacuole symbiotically infected by bacteria (s.s., bacteriology)specialized compartment in a host cell that houses an endosymbiont (s.l.) |
Domain Archaea (始原菌, 古細菌, アーケア)Phylum CrenarchaeotaClass Thermoprotei Reysenbach 2002Order Thermoproteales Zillig et Stetter 1982ThermoproteaceaeAnaerobic thermophilic sulfur archaea (嫌気性好熱硫黄古細菌): Thermoproteus, Caldiviga, Pyrobaculum (grown well ≈ 100°C), Thermocladium Thermofilaceae Thermofilum Order DesulfurococcalesDesulfurococcaceaePyrodictiaceae Order SulfolobalesPhylum Euryarchaeota Garrity et Holt 2002Class Methanobacteria Boone 2002methane bacteriaOrder MethanobacterialesMethanobacteriaceae: Methanobacterium, Methanobrevibacter, Methanosphaera, MethanothermobacterMethanothermaceae: Methanothermus Class Methanococcimethane producerClass Halobacteriaextreme halophile, reproducing under high NaCl concentration |
Class Thermoplasmataacidophiles, growing optimally at pH below 2Class Thermococcigrwoing in extremely hot environmentsClass Archaeoglobi Garrity et Holt 2002three genera in one family, grwoing in extremely hot environmentsArchaeoglobaceae: Archaeoglobus, Ferroglobus, Geoglobus Class MethanopyriOne species:Methanopyrus Kurr et al. 1992: M. kandleri Kurr et al. Environments (環境)1) Temperaturepsychrophile (好冷生物), psychrophilic bacteria (好冷菌): < 15°Cmesophile (好中温生物): 15-40°C thermophile (好熱生物), tempmophilic bacteria (好熱菌): 45-80°C hyperthermophile 超好熱生物: > 80°C 2) pHacidophile (好酸性生物): < 5neutrophile (好中性生物): 6-8 alkaliphile (好アルカリ性生物/菌): > 8 3) Salt (halophile)haplophilic bacteria (好塩菌)4) PressureMethanogen (メタン菌)= methane-producing bacteriaMethanobacterium, etc. 4H2 + CO2 → CH4 + 2H2O |
⊂ Domain Eukaryota (Eukaryotes)(Chatton 1925) Whittaker et Margulis 1978Revisions to the classification, nomenclature, and diversity of eukaryotes (Adl et al. 2019) Kingdom (or unranked) AmoebozoaLühe 1913 sensu Cavalier-Smith 1998= Eumycetozoa Zopf 1884, emend Olive 1975 Infraphylum Mycetozoa de Bary 1873 (粘菌類)= a polyphyletic grouping of slime moldsSlime mold or mould: has a life cycle that includes a free-living single-celled stage and the formation of spores Class Protostelea (原生粘菌), protosteloid amoebae
= protosteloids, protostelids |
Class Dictyostelia Lister 1909, emend. Olive 1970 (タマホコリ), dictyostelids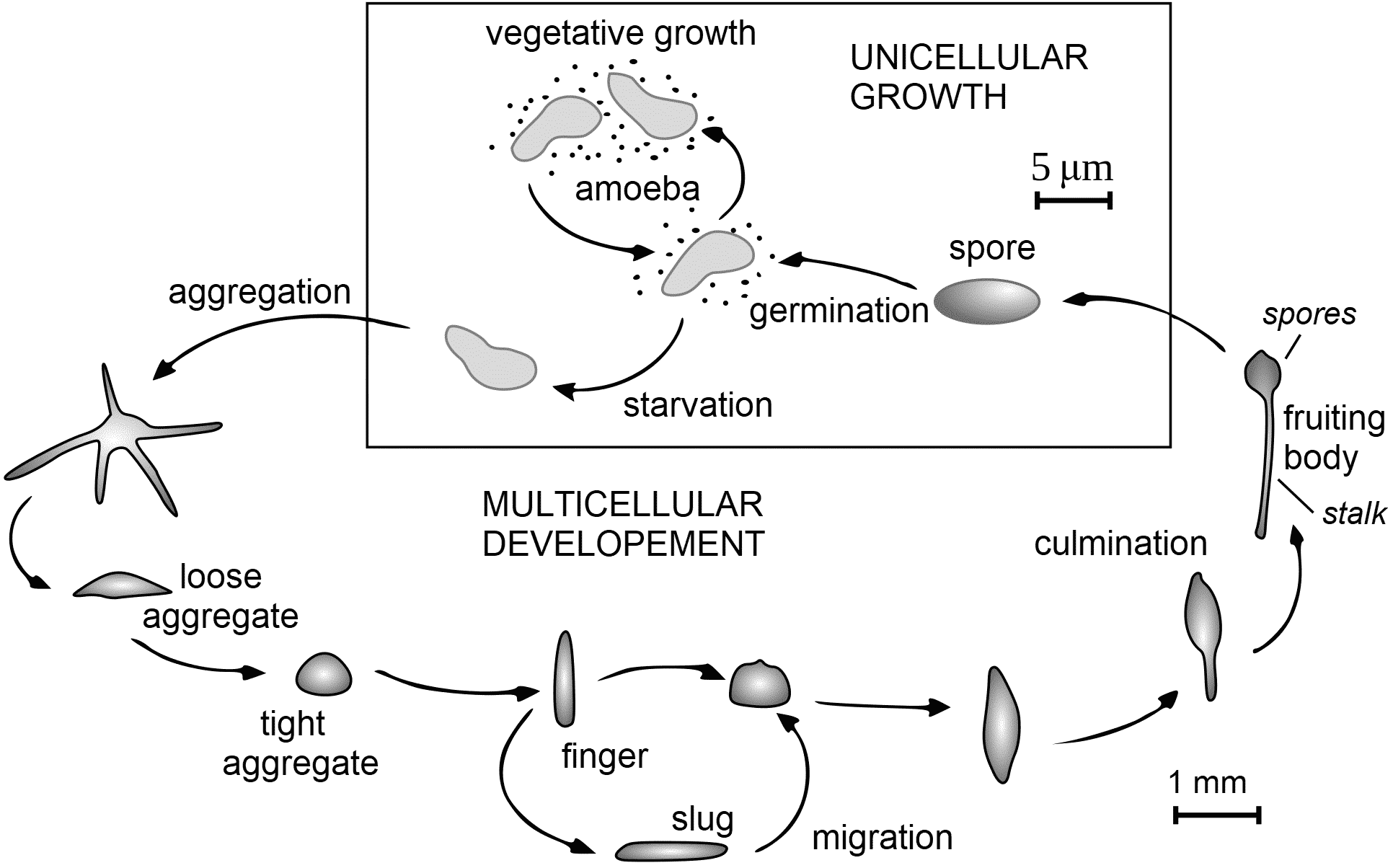 Fig. Life cycle of Dictyostelium. Aggregate (aggregation, 細胞凝集). Fruiting body (子実体). Culmination (子実体形成). (Macro)cyst (spore, シスト) Order Dictyosteliida or Dictyosteliales (タマホコリ)= dictyostelids, Dictyostelea (ICZN) or Dictyosteliomycetes (ICBN)(dictyostelid) cellular slime molds Dictyosteliidae or Dictyosteliaceae Rostafinski (タマホコリ) Dictyostelium Brefeld (タマホコリ) D. discoideum Raper (キイロタマホコリ): model organism D. purpureum Olive (ムラサキタマホコリ) D. minutum Raper (コタマホコリ) Polysphondylium Olive (ムラサキホコリモドキ) P. violaceum Brefeld ムラサキカビモドキ P. pallidum Olive シロカビモドキ Coenonia Myxomitodes, fossil record Actyosteliidae (エツキタマホコリ) = Acytosteliaceae Raper ex Raper et Quinlan 1958 Acytostelium Raper (エツキタマホコリ) |
Gram stain or Gram staining (グラム染色)Note that young cells can not be stained wellFour basic steps:
|
 methyl violet 10B = crystal violet (6)___MV 6B (5)___________MV 2B (4) |
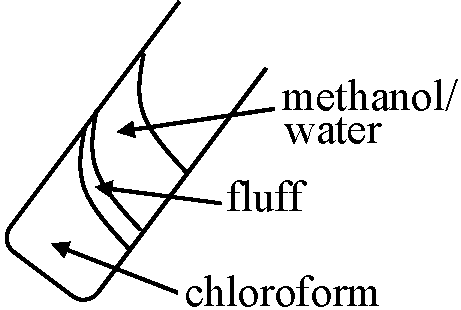
Extraction procedurePreculture Pseudomonas aeruginosa at 20°C for 48 hrMain culture (nutrient medium 1% peptone) : transfer 4 ml pre-culture 4 ml
at 20°C for 21 hr Wash suspension (WW ca 2g/20 ml)
↓ add 50 ml methanol |
Wait for 60 min (do not forget this!) ↓ add 25 ml chloroform Rigorous mixing for 15 min
→ add 15 ml 10mM NaCl Add 20 ml chloroform to the remainder* (*: repeat these for 3 times) Recover chloroform layer and centrifuge Recover chloroform gently |