(Upload on September 29 2025) [ 日本語 | English ]
Mount Usu / Sarobetsu post-mined peatland
From left: Crater basin in 1986 and 2006. Cottongrass / Daylily
HOME > Lecture catalog / Research summary > Glossary > Biological and physical cycle
geosphere (地圏)domain of the earth inside
↓ lithosphere hydrosphere (水圏)domain consisting of water ≈ ocean + lake + riveratmosphere (大気圏)domain covered with airbiosphere (生物圏)domain where natural life forms occur (s.l.) → biota (s.s.) |
[biogeochemical cycle, nitrogen cycle, water cycle, biomagnification, trophic level, photosynthesis] Material and energy cyclesBalance of energyOne of Commoner's lasting legacies is his four laws of ecology, as written in The Closing Circle in 1971. The four laws are:
|
|
Circulation at global scale → material cycle observed between atmosphere-hydrosphere-geosphere-biosphere
thermal cycling water cycle = hydrologic cycle atmospheric circulation thermohaline circulation material circulation, etc. Classification by materialsNitrogen cycle (N) (窒素循環)Oxygen cycle (O) Carbon cycle (C) (炭素循環) |
Phosphorus cycle (P) (リン循環)
P stocks = soil water, rivers, lakes and rocks along with ocean sediments
Three processes release S to the atmosphere Hydrogen cycle (H), etc. |
| Stemflow (樹幹流) | |
|---|---|
|
Def. the flow of intercepted rainwater that travels down the trunk or stem of a plant
a mechanism for concentrating water and solutes at the base of trees, influencing soil moisture, nutrient distribution and ecological processes |
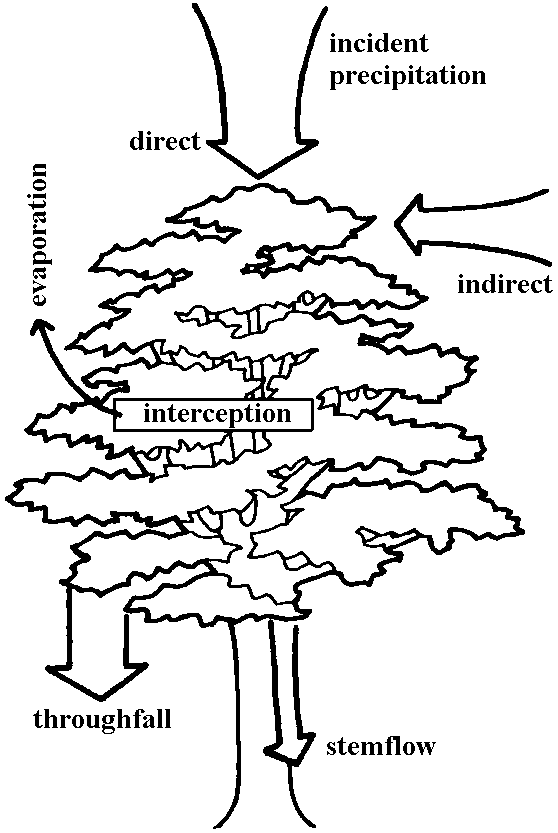 (Parker 1983) |
Detecting: direct linkage between lithosphere, biosphere, atmosphere, etc.
StandardSample ratio as it relates to the standard is in the following form:δX (‰) = (Rsample - Rstd)/Rstd × 1000 ______ = (Rsample/Rstd - 1) × 1000
δ: the isotopic notation
Δ = {(δsource - δsample)/(1 + δsample/103)} × 103 or International standardsA 0‰ value of the δ-scaleInternal lab standards must be corrected to international reference standards δ13C = Vienna peedee belemnite (VPDB) δ15N = atmospheric nitrogen Fractionation (分別)Equilibrium fractionation, e.g.,α18Owater-vapor = (18O/16Owater/18O/16Ovapor)
occurring when substrates and products of chemical equilibrium reactions differ in their isotope ratios because the heavier isotopes create stronger bonds with either the substrate or product α = Rreactant/Rproduct
occurring when a single type of molecule changes phase (e.g., from liquid to gas) or when the chemical reaction is non-reversible 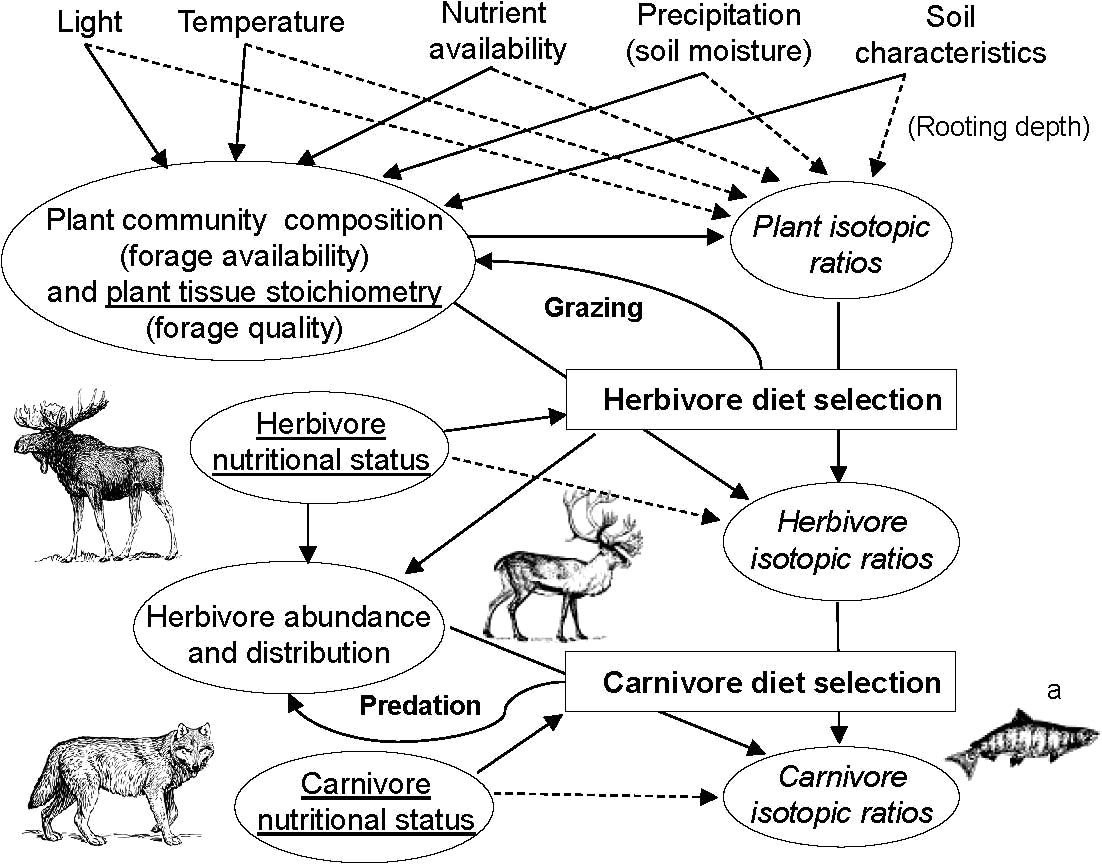 |
Fig. Interactions between processes that influence stable isotope ratios of herbivores and carnivores, showing biochemical, physiological (underlined), and behavioral (in rectangles) processes. Solid lines represent ecological interactions; dotted lines represent factors affecting diffusion rates and enzymatic reactions (i.e., photosynthesis, nutrient routing and nutrient recycling). A single isotopic value obtained from tissue of a carnivore is the emergent property of multiple ecological, behavioral, and physiological processes of various ecosystem components. (Modified from Ben-David et al. 2001). (a) Effects of marine subsidies on wolf diets and ungulate population dynamics are described in Adams et al. (2010). Fig. Trophic enrichment in δ13C and δ15N from primary producers (plants), to herbivores, to predators for terrestrial ecosystems. The panel also shows differences in δ13C between food webs based on C3 (black symbols) and C4 plants (gray symbols). Values (mean ± SE) are adapted from the following sources: willows (Salix) from Ben-David et al. (2001); moose (Alces alces) and wolves (Canis lupus) from Szepanski et al. (1999); grasses from Wang et al. (2010); zebra (Equus burchellii) and lions (Panthera leo) from Codron et al. (2007). (Ben-David & Flaherty 2012) Plant samplesMeasurement: TCEA-IRMS (isotope-ratio mass spectrometry)Sample: dried at 60-80°C → grinded the samples at 40 μm The smaples are stable after drying up |
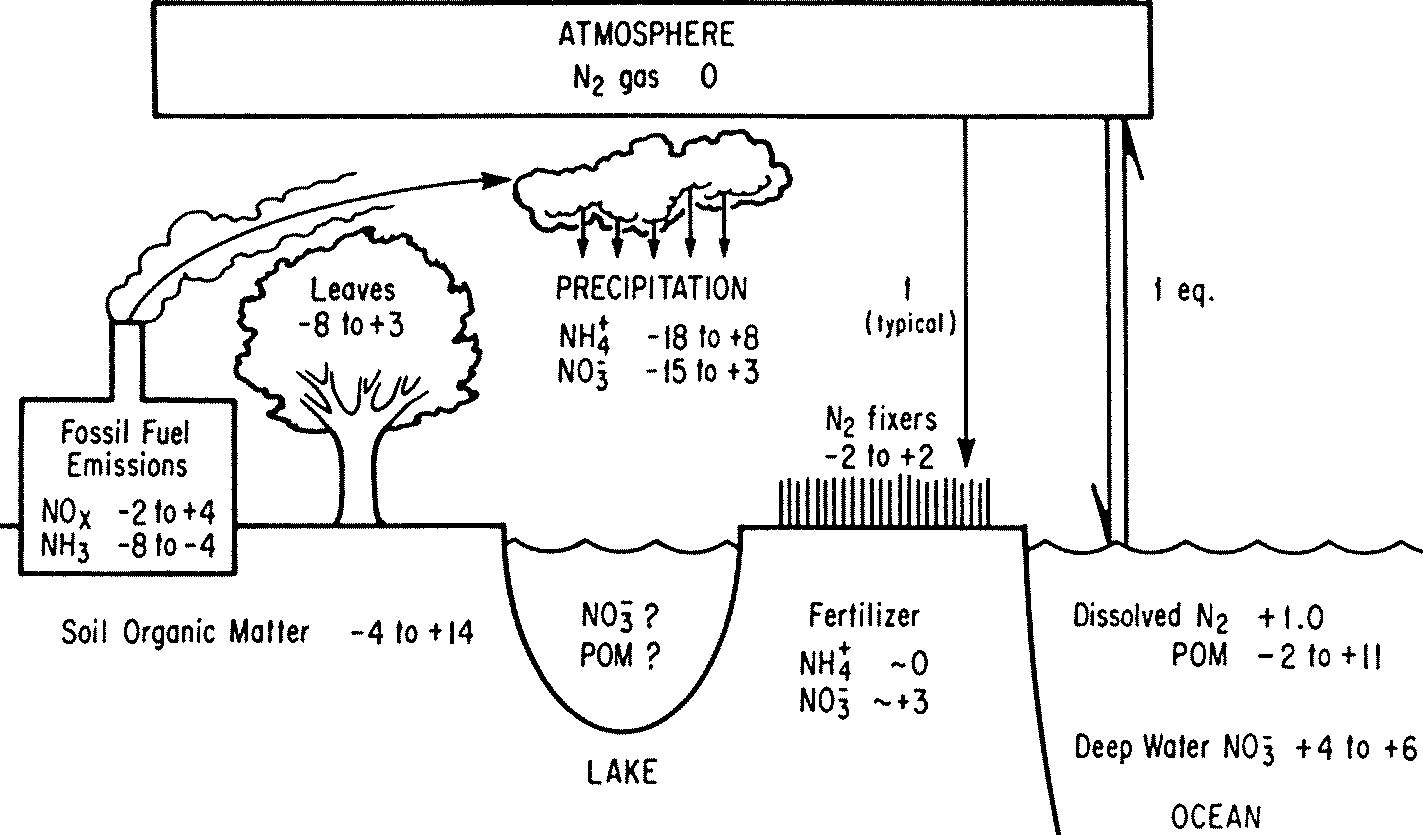 Fig. δ15N in an ecosystem. The unit of numeral is ‰.
Atmospheric reservoir of 0‰ 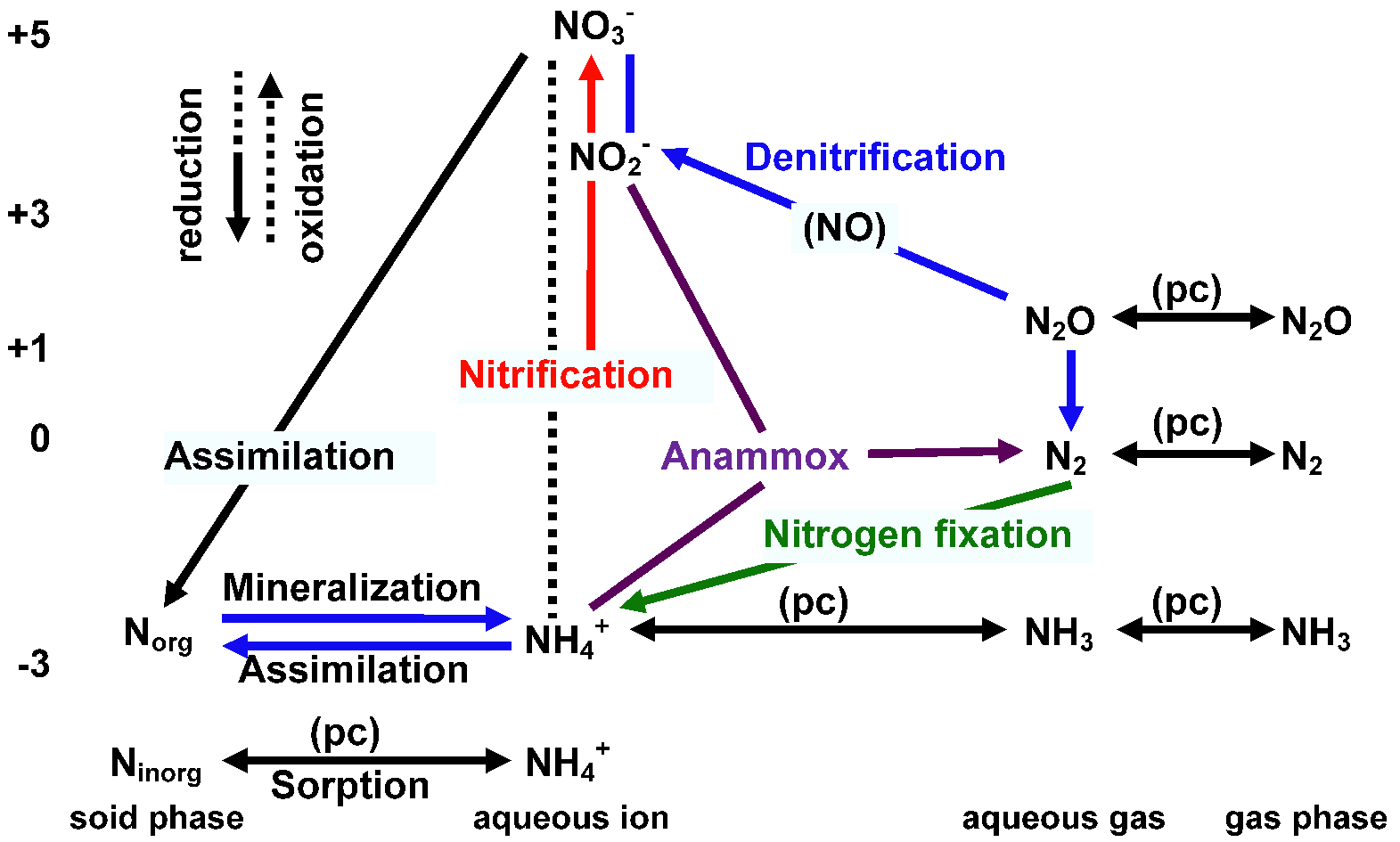 Fig.1. Biogeochemical and physical-chemical (pc) processes affecting the speciation of nitrogen in aquatic systems. Highlighted are some of the major reactions considered in the current study, including nitrification, denitrification, anammox, and NH4+ exchange with solids assimilation (同化), anammox (anaerobic ammonium oxidation) (嫌気性アンモニア酸化), denitrification (脱窒), mineralization (無機化), nitrification (硝化, 硝化作用), nitrogen fixation (窒素固定) Norg: proteins of plants, animals and microbes, etc. A. Nitrogen fixation (窒素固定)(1) Physical or chemical nitrogen fixation(2) Biological nitrogen fixation: nitrogen fixing bacteria (窒素固定菌) Extracellular symbiont azotobacter around plant roots, not developing specific organs anabaena: fern developing specific leaves for anabaena |
Intracellular symbiont Rhizobium (根粒菌): rrhizobium = root nodule bacterium - polyphyletic
specific host = leguminous plants
approximation, by the chemotaxis of bacteria
moving to cortical cells + bacteroid: nif gene group = all genes related to nitrogen fixation
nif genes coded by plasmid in the bacteria ≈ nitrogen fixer, Klebsiella pneumoniae nodulin: nodule-specific protein, function unknownleghemoglobin (leguminous hemoglobin)
structure and function comparable with hemoglobin → Nitrogen-fixing bacteria that forms symbiotic relationships with actinorhizal plants initiating the formation of root nodules, where they convert atmospheric nitrogen into ammonia Hosts
all Alnus species (Betulaceae) B. Nitrogen assimilation (窒素同化)NH3 produced by isolated root nodulde bacteria under specific conditions→ nitrogen fixation is conduced by the bacteria C. Denitrification (脱窒)Microbially facilitated process of whcih nitrate (NO3-) is reduced and finally produces N2Half reactions
NO3− + 2H+ + 2e− → NO2− + H2O (nitrate reductase) 2NO3− + 10e− + 12H+ → N2 + 6H2O Nitrate reduction (硝酸還元)Nitrite reduction (亜硝酸還元) |
|
Terrestrial plants: use atomospheric CO2 Aquatic plants: use dissolved carbonates (hydrosphere carbon)
Terrestrial Oceanic |
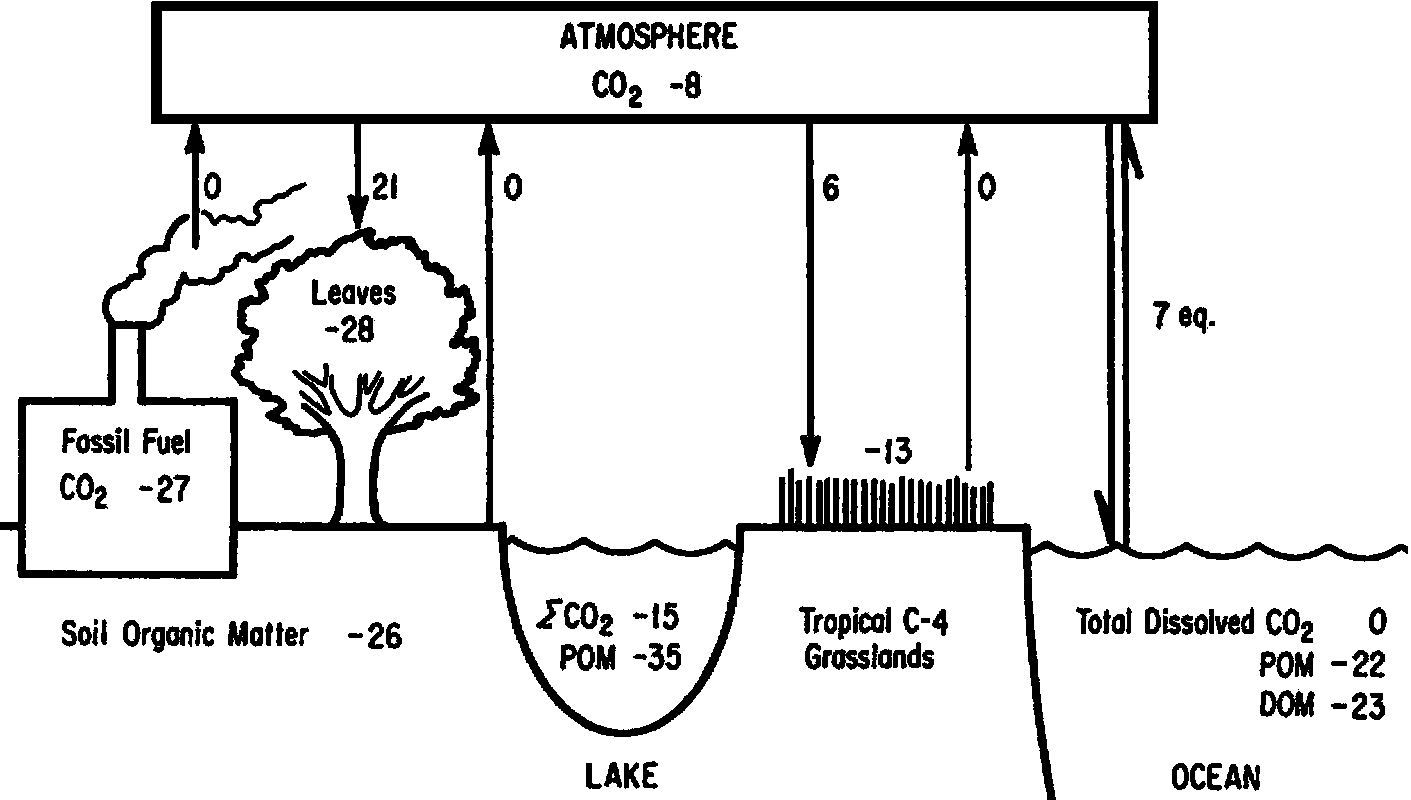 Fig. δ13C in an ecosystem. The unit of numeral is ‰.
Atmospheric CO2 (≈ -8‰) |
Bioaccumulation (生体蓄積・生物蓄積・生物濃縮)= biological accumulationthe biological sequestering of a substance at a higher concentration than that at which it occurs in the surrounding environment or medium → biomagnification + bioconcentration Biomagnification (生態濃縮)= bioamplification or biological magnificationthe increase in concentration of a substance, such as a pestside, in organisms along a food chain or web as a consequence of: |
persistence (not broken down by environmental processes) Bioconcentration (生物濃縮)= biological concentrationthe accumulation of a chemical in the tissues of an organism as a result of direct exposure to the surrounding medium, such as water (i.e., it does not include food web transfer) The three terms described above are often jumbled up. |
|
Seagrass is recorded from two (Engler's syllabus エングラー体系) to six families with ca 60 species: All of them are in monocotyledons Potamogetonaceae (ヒルムシロ)
Zosteraceae (アマモ) |
Cymodoceaceae (ベニアマモ) Hydrocharitaceae (トチカガミ) |
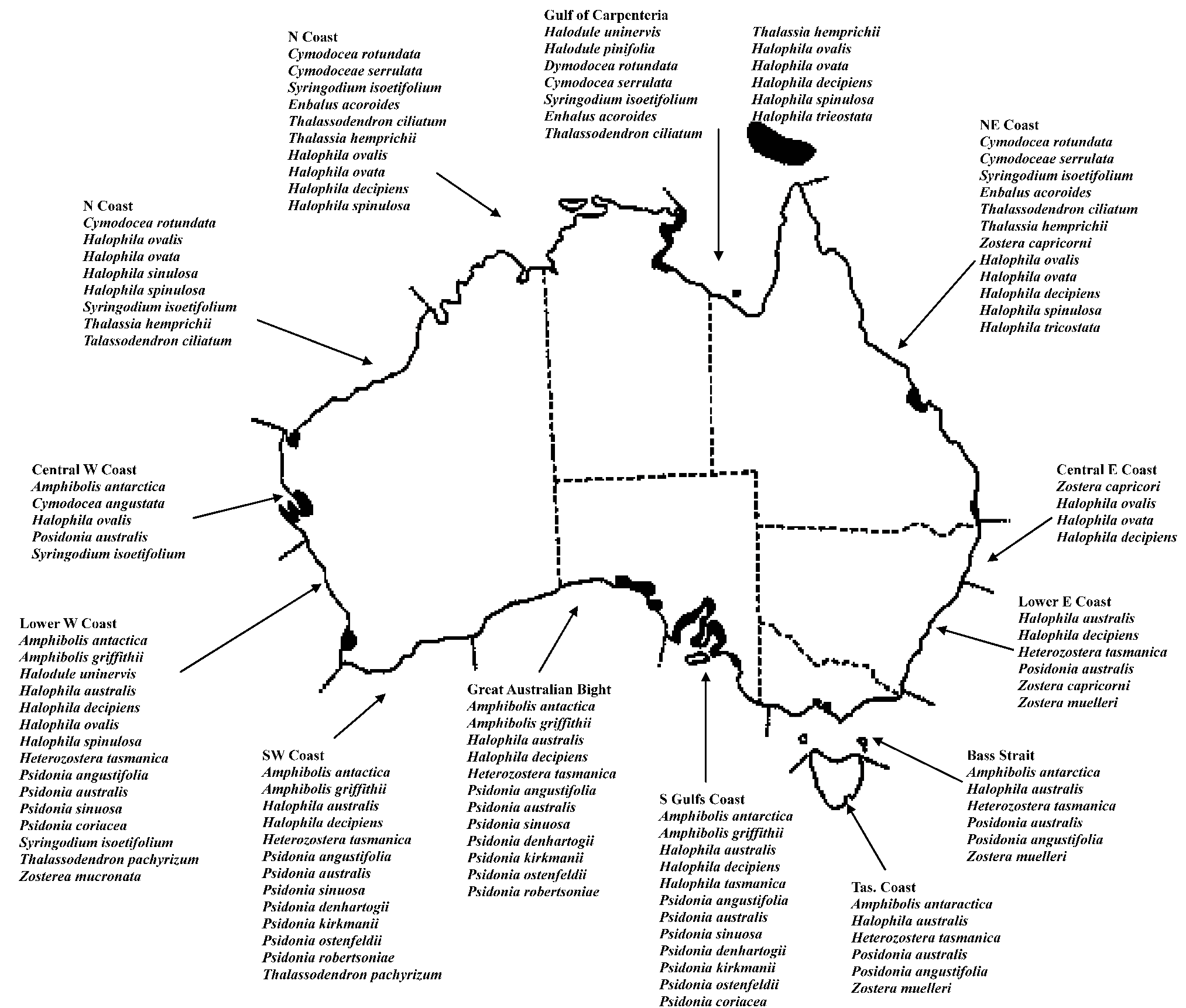
Fig. 1. Distribution of seagrass species along the Australian coast line divided into CONCOM regions.
Seagrass meadows are the shaded areas along the coast and the lists are of seagrass species found in each CONCOM region.
Research on ocean ecosystems and the interactions between chemical cycles and life
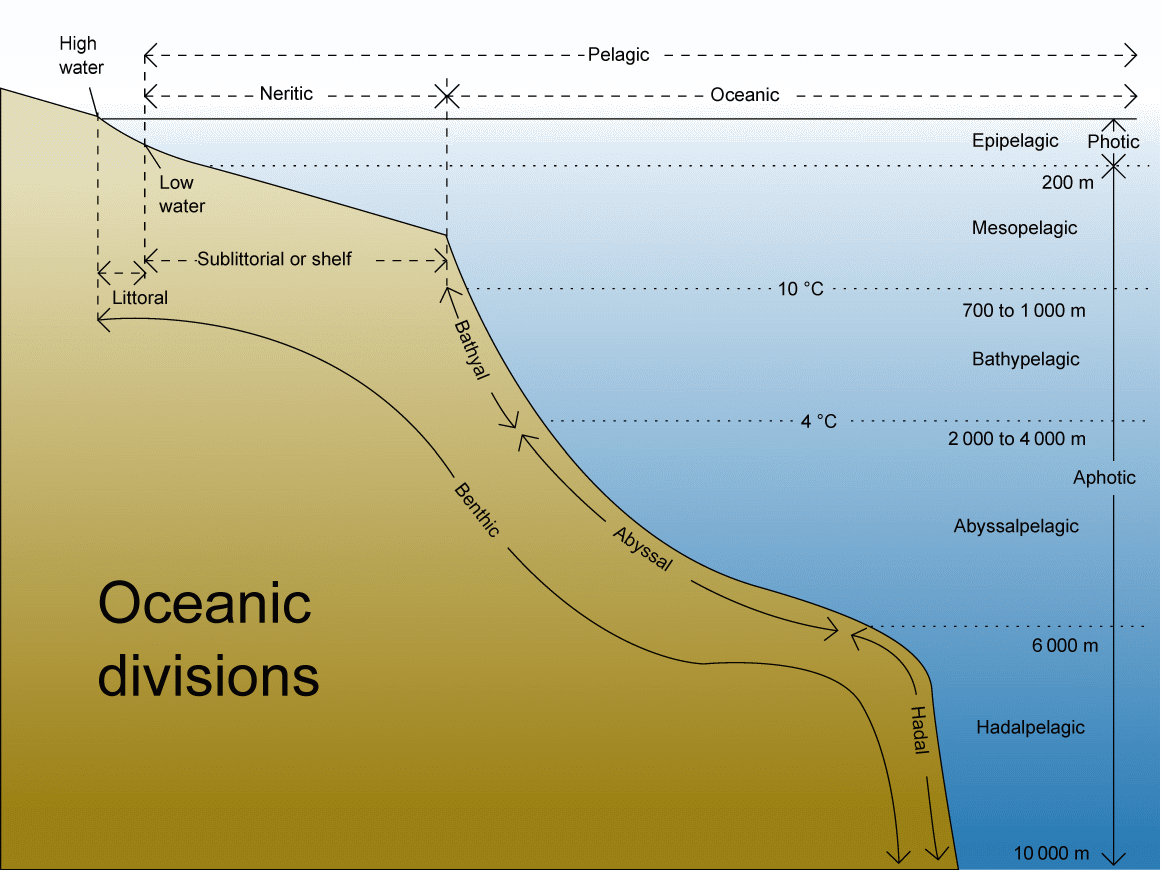 Pelagic zone (漂泳帯): water column where swimming and floating organisms live
= neritic + oceanic zones ≈ Epipelagic zone (海面表層) Aphotic zone (無光層)
Mesopelagic (中深層) Light and vertical distributionLambert-Beer law (ランバート・ベーア則)= Beer-Lambert law, Beer-Lambert-Bouguer law, or Beer's lawShowing the abosrbance curves of light intensity with increasing the depth of water or other materials (in particular in sea) Application: chemical measurements I = I0·e-KD
D: depth (or leaf area index above the depth) K is obtained by I/I0 and D |
Photosynthetic productivity, Qx = ∫P(λ)·Ix(λ)·dλ
x: depth 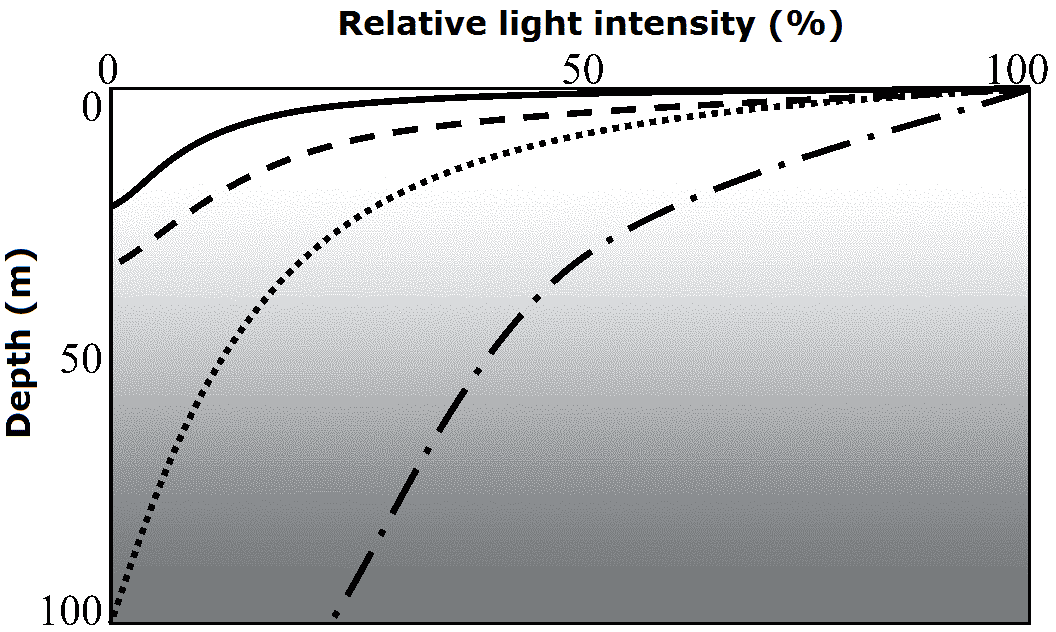 Sea surface temperature, SST (海面温度)Paleothermometry (古温度測定法)TEX86 paleothermometry Analisysi of organic molecules by LC-MS/MS Euphotic zone (有光層)Layers of which photosynthesis is higher than respiration → plants can grow= range beteween water surface and compensation depth (補償深度)
Marine: compensation depth (m) Littoral zone (沿岸帯)Zonation: three zones are developmed in most littoral communities↓ Supratidal zone (supralittoral zone) ↓ Eulittoral zone Intertidal zone (tidal zone): occurring between the levels of low and high tides; thus, exposed at low tide; also reffered to as eulittoral ↓ Sublittoral zone Tide pool (us → en = rock pool)developing specific ecosystems Aquatic organisms (水生生物)lifeform: determined mostly by the movement of water (water flow) Plankton: the community of minute organisms suspended in waterMeroplankton (meroplanktonic, adj.): refers to organisms that spend a part of their life cycle as planktonic and a part as bentic (as in a resting stage), which restricts such organisms largely to neritic conditions Necton (or nekton): marine and freshwater organisms that swim freely and are generally independent of currents, ranging in size from microscopic organisms to whales Ex. fish, squid, octopusBenthos: organisms which live on, in, or near the seafloor (called benthic zone). Ex. bentic algae, barnacle, sea urchin, sea cucumber + Seston: the organisms (bioseston) and non-living matter (abioseston or tripton) swimming or floating in a water body. → seeds of seagrass |
| Environmental issues (環境問題) | |
|---|---|
Bloom (異常発生)Harmful algal bloom (HAB), e.g., red tideRed tide (赤潮)= red water, or discolored watera phenomenon where colonies of algae grow out of control in marine or freshwater environments, often producing (neuro-)toxins that negatively affect marine life, birds and even humans |
Causes: nutrient loading, temperature rise, water column stratification and ocean currents Algal species: often dinoflagellates, such as Karenia brevis, Alexandrium, Gymnodinium each with different toxins and behaviors Blue tide (青潮)Two different uses:
1. Upwelling of hypoxic or anoxic water in coastal seas |