(Upload on November 12 2017) [ 日本語 | English ]
Mount Usu / Sarobetsu post-mined peatland
From left: Crater basin in 1986 and 2006. Cottongrass / Daylily
HOME > Lecture catalog / Research summary > Glossary > Seed germination
|
Def. germination; the development of a plant from a seed or spore after a period of dormancy e.g., seed germination
Def. emergence: the process of becoming visible after being concealed Ex. seedling emergence seed → germination → seedling
seed dispersal → Germination signalTwo signals: dormancy-break signal + germination signal → germinationGermination: The initiation of embryo growth and seedling development. Dormant seed: Viable seed in a non-germinable condition due to internal and/or external barriers, e.g., hard seed coat, water defficiency, and no light. 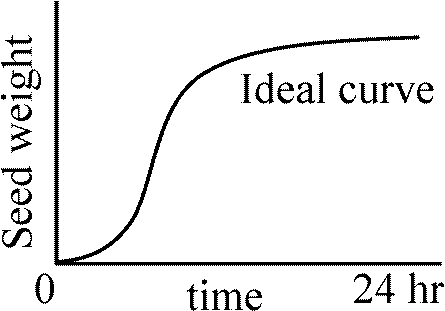
Seed imbibitionMeasurement of seed imbibition rate - seed imbibition (rate) curve5 lots of 10 seeds × 9 replications = 45 lots Therefore, you need more than 450 seeds for 1 species Measure seed weights at 3 hr intervals until no more seed weight increase is observed |
Warm stratificationAny seeds that are indicated as needing a period of warm stratification followed by cold stratification should be subjected to the same measures, but the seeds should additionally be stratified in a warm area first, followed by the cold period in a refrigerator later. Warm stratification requires temperatures of 15-20°C. In many instances, warm stratification followed by cold stratification requirements can also be met by planting the seeds in summer in a mulched bed for expected germination the following spring. Some seeds may not germinate until the second spring.Heat exposure (熱)Dry heat:In an oven at 75°C for 25 min, using 40 species (Tsuyuzaki & Miyoshi 2009) In a water bath, ranging from 35°C to 95°C for 30 min, for three Asteraceous species (Yuan & Wen 2018)
Dry heat: without pouring water in the flask |
Germination indicesGermination percentage (germinability), gpgp = Ng/Nt × 100 (%)
Ng = the number of germinated seeds Time for the first germination or germination time lag, t0the time for first germination observed (unit: time, e.g., day)Time for the last germination, tgthe time for last germination observed (time)Time spread of germination, or germination distribution, TsTs = Tg - T0 (time)Peak period or modal time of germinationthe time showing the highest frequency of germinated seeds (time)Median germination time, t501. Coolbear et al. (1984), t50 = Ti + [{(N + 1)/2}(Tj - Ti)]/(Nj - Ni) (time)
t50: median germination time Mean (Ta), variance (s2T) and standard error (sT) of germination timemean germination time = mean length of incubation time, germination resistance or sprouting indexTa = Σi=1kNiTi/Σi=1kNi (time)
Ti: the time from the start of experiment sT = √(s2T/Σi=1kNi) (time) Mean germination rate (Va), variance (s2V) and standard error (sVa)Va = Σi=1kNi/Σi=1kNiTi = 1/Ta (time-1)s2V = Va4·s2T (time-2) sVa = √(s2v/Σi=1kNi) (time-1) Coefficient of germination velocity, Cgv= Coefficient of germination rate (Cgr) or Kotowski's coefficient of velocityCgv = (Σi=1kNi/Σi=1kNiTi)·100 = Va·100 (% day-1) Germination rate as the reciprocal of the median time, v50v50 = 1/t50 (time-1)Speed of germination, S= germination or emergence rate index, or index of germination velocityS = N1/T1 + N2/T2 + N3/T3 + … + Nn/Tn = Σi=1n(Ni/Ti) (% time-1) Speed of accumulated germination, SaSa = (N1/T1) + Σi=12(Ni/T2) + Σi=13(Ni/T3) + … + Σi=1n(Ni/Tn) (% time-1)Corrected germination rate index, ScSc = S/gp (time-1)Weighted germination %, gwgw = Σi=1t{(t - i + 1)Ni/(t·N)}·100Mean germination % per unit time, gugu = gp/TnNumber of seeds germinated per unit time, gNgN = Ng/TnTimson's index, T, or germination energy indexT = Σn = Σi=1tGi = Σi=1tgi(t - j), j = i - 1
Gi: the cumulative germination % in time interval i Modified Timson's index, Tm1. Labouriau, Tm = T/Σi=1tgi2. Khan and Unger, Tm = T/t George's index, GrGr = Σi=1tNiKiKi: the number of intervals until the end of the test Peak value (PV) or emergence energy (EE)the accumulated number of seeds germinated at the point on the germination curve at which the rate of germination starts to decrease |
Germination value, gv1. Czabator, gv = pv·gmdgmd: the mean daily germination % from the onset of germination (computed for other time intervals of successive germination counts, by replacing gmd with gp) 2. Diavanshir & Pourbiek, gv = (Σgds)/N·go·k
gds: the temoporal germination speed by dividing cumulative germination % by the number of days since the onset of germination Coefficient of germination uniformity, gucguc = (Σi=1kNi)/(Σi=1k(Ta - Ti)2Ni)Coefficient of germination time variation, gtcgtc = √(s2T/T)Synchronization index, Ea= uncertainty of the germination process, U, or informational entropy, HEa = -Σi=1kfilog2fi fi: relative frequency of germination = Ni/Σi=1kNi Germination synchrony (Z index)Z = (Σi=1kCNi,2/CΣNi,2)
CNi,2 = (Ni(Ni - 1))/2 (Gummerson 1986) Hydrothermal time model (HTT model)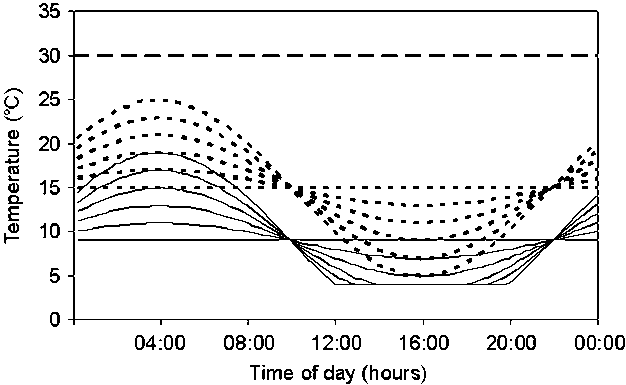 Fig. 1. Diurnal temperature courses as applied in the 13 different temperature regimes in Experiment 2 for germination tests of Solanum sisymbriifolium seeds. Separate temperature regimes are indicated by numbers in the rest of the manuscript: solid lines represent regime 1, which was a constant 9°C treatment. Regimes 2, 3, 4, 5, 6 were treatments with fluctuating temperatures around 9°C with maximum amplitudes of 2, 4, 6, 8, and 10°C, respectively. Dotted lines represent regime 7, which was a constant 15°C treatment, and regimes 8, 9, 10, 11, 12, which were treatments that fluctuate around 15°C with maximum amplitudes of 2, 4, 6, 8 and 10°C, respectively. The broken line is regime 13, a constant 30°C treatment. Fluctuations were sine functions (of wavelength 1 d) with daily means of 9°C (treatments 2-6) and 15°C (treatments 8-12), respectively. The functions were truncated at 4°C, thus avoiding potential damage due to very low temperatures. (Timmermans et al. 1997) ____Rumex obtusifolius___Polygonum longisetum___Oenothera biennis 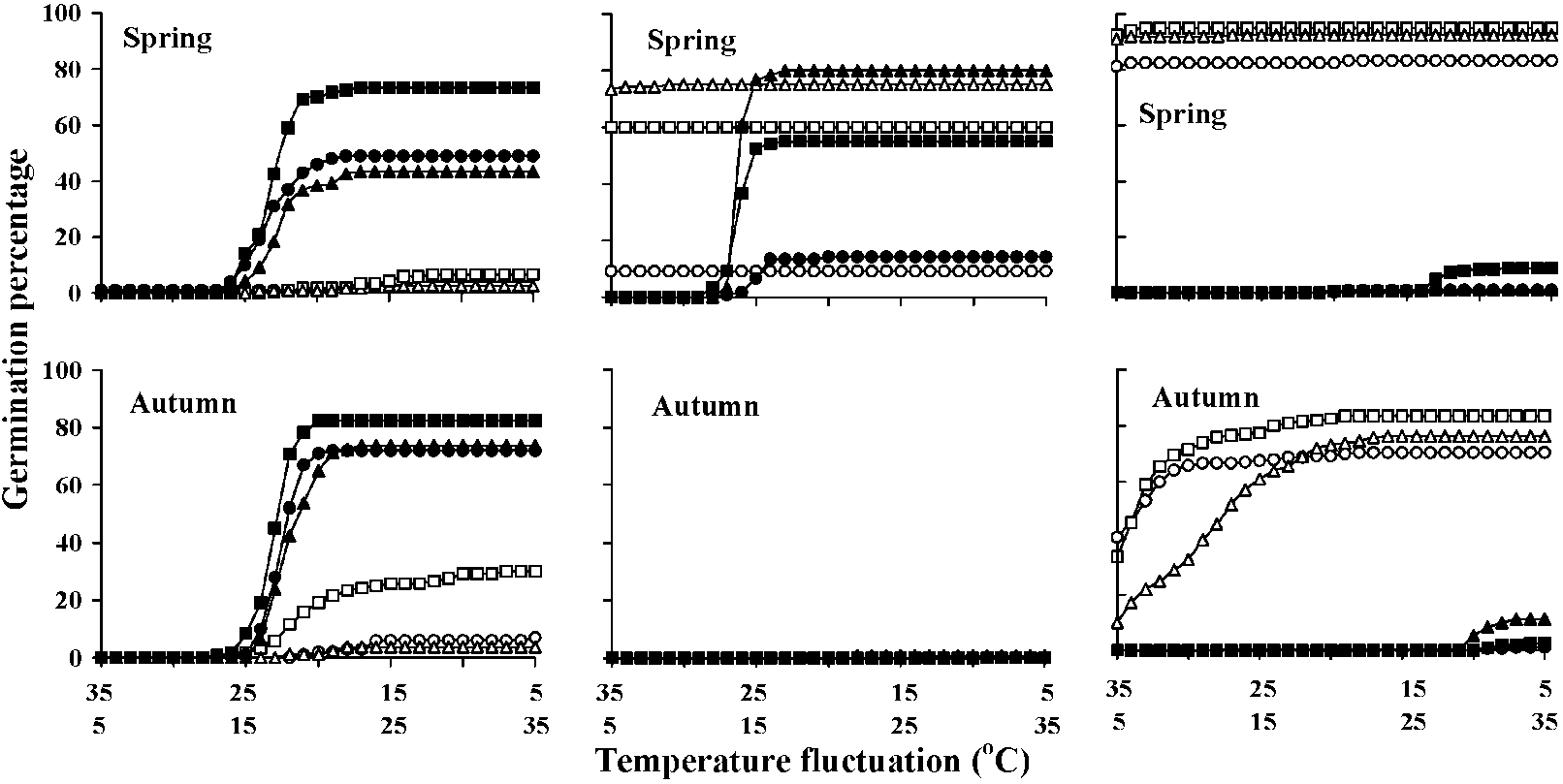 Fig. 1. Seed germination curves for three species buried at three soil depths (3, 10 and 30 cm). Mean germination percentage on six replicates was shown. All standard errors are less than 4 and are not shown in the figure, Closed symbols indicate temperature changes from low to high (L-H) and open symbols from high to low (H-L). Circles, squares and triangles are for seeds buried at depths of 3, 10 and 30 cm, respectively (Tsuyuzaki 2006) |
Pre-treatment
SterilizationTo reduce the damages of seeds by fugui (and bacteria), sterlization may be required. We usually use benlate or 1% sodium hypochlorite (NaClO, bleach).Cold stratification (低温湿層処理)To increase the seed germination rate, cold stratification is required for many species that are distributed in cool and temperate regions. Protocolkeep the seeds in 0-6°C for 1 month with moisture
moistened on three-layered filter papers in a petri dish (proposed by Suzuki S) Daily temperature fluctuationDaily temperature fluctuations during seed germination testA) Treatment under low temperature = simulation of fall condition
light: light/dark = 8 hr/16 hr with more than 1000 lux
light: light/dark = 8 hr/16 hr |
Exp. Germination characteristics of Gaultheria miqueliana seeds
Preparation: 40 petri dishes, 120 filter papers (Whatman #1), dim green light, distilled water, twizzers, wrap H2SO4 soaking or sand scratchProblems1) concentration of sulfuric acids 2) treatment time → need preliminary survey
1) H2SO4 treatment (only): one sample = 50 seeds, time: 1 min, 2 min, 5 min, 10 min, 20 min, 40 min. GA3 treatmentoften using to promote the seed germination of buried seeds
concentration of GA3: 0 (control), 100, 200, 400 ppm
↓ Soaking seeds into condensed H2SO4 solution within 1 minute |
Greenhouse (温室)


Seed germination exeriment conducted in a greenhouse. The samples were collected from the former topsoil on Mount Usu 20 [left] and 30 [right] years after the 1977-78 eruptions. The most common species was Rumex obtusifolius (Left: Goto M. Right: ST). |
A few photos on seedsJuncus effusus var. decipiensPolygonum weyrichii Phytotron (ファイトトロン)A closed system that regulates various environments used for investigating plant growthLED light PRO180 SODATEC (out of order)
PAR (μmol): 1469.2 |
[ dromancy ]
|
A period in a life cycle when seed development is temporarily suspended Innate dormancy (Primary dormancy)seed is dormant during seed maturation (true dormancy)After-riping: seeds requried a period to be mature after the dispersal Induced or spontaneous dormancy (seconcary dormancy, 自発休眠)seed does not germinate spontaneously until environmental conditions improve. The seed needs favorable environmental stimuli to germinateEnforced or imposed dormancy (quiescence, 強制休眠)seed germinates readily on removal of the environmental limitationConditionswaterlight temperature gasses (O2, CO2, etc.) smoke mechanical restriction seed coat hormone |
(Baskin & Baskin 2003) Exogenous dormancycaused by conditions outside the embryo
Endogenous dormancycaused by conditions within the embryo itself
Stagepre-dormancy (前休眠) or summer dormacny (夏休眠) →
true or winter dormancy (冬休眠) at innate dormancy stage → |
Water stressWhen water demand exceeds the available amount, the various changes are observed with various sclales from molecule to ecosystem. Water stress on seed germinationWhen water is deficient, seed germination percentage often decreases to avoid drought stress on seedlings. Protocolto detect the effects of water stress on seed germinationMethod 1 (classical method)Changes in the thickness of seed bed by adjustijng nunmber of filter peaprs, and keeping constant water table by adjusting water supplyMethod 2PreparationMaking an osmotic pressure gradient by the concentration of polyethylene glycol (PEG) |
Equation on making osmotic perssure using PEG-6000ψs = -(1.18 × 10-2)C - (1.18 × 10-4)C² + (2.67 × 10-4)CT + (8.39 × 10-7)C2T
ψs: osmotic pressure (bar) Polyethylene glycol (PEG)≈ polyethylene oxide (PEO), and polyoxyethylene (POE)PEG: tended to refer to oligomers and polymers with a molecular mass < 20000 g/mol PEO: tended to polymers with a molecular mass > 20000 g/mol POE: tended to a polymer of any molecular mass. |