(Upload on October 4 2021) [ 日本語 | English ]
Mount Usu / Sarobetsu post-mined peatland
From left: Crater basin in 1986 and 2006. Cottongrass / Daylily
HOME > Lecture catalog / Research summary > Glossary > Water
Water for life (生命にとっての水)Plants live with water because of photosynthesis (光合成)
Plants [ algae | moss | fern | seed plant ] Water types used by plants1. rainfall (降雨)Worldwide average: 880 mm/yr - highly heterogenous distribution 2. stream water (河川水) |
[ water potential | pH ] phreatophyte (地下水植物): plants utilizing groundwater Ex. Poenix dactylifera (ナツメヤシ) - developing oasis, Prosopis juliflora (メスキート) Hydraulic redistribution 4. mist and dewcloud forest or mist forest - how to measure! |
Density (密度), ρTemperature-density relationship (温度-密度式)(1) proposed by Eddington, Sir Arthur (Archibald) Stanley (1882-1944) ρ(T) = ρ0[1 - β(T - T0)]
ρ(T): the density at temperature T Note that this equation is simplified formula |
(2) a precise formula, when required (Kell & Chera 1975)
0 ≤ T (°C) ≤ 100 ⇒
-46.170461 × 10-6T3 + 105.56302 × 10-9T4 at 0°C = 0.9998 3.95°C = 0.999972 100°C = 0.9584 (> ice) |
|
(Slatyer & Taylor 1960) Water potential (水ポテンシャル)Quantifier: ψ, or ψwDimension: L-1MT-2 Unit: Pa
Water transport: from high to low water potential →
ψl < ψx < ψr < ψs ψh = ψm + ψg (Pa or cm)
ψh: hydraulic potential (water potential) 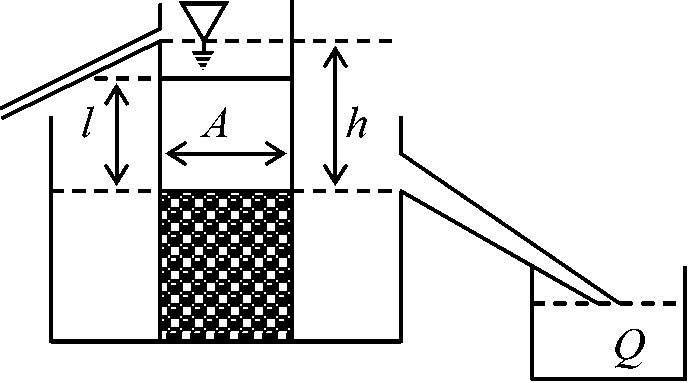 J = -K·(dh/ds) (m3/m2s = m/s)
J = -K·(dh/ds) (m3/m2s = m/s)
J: flux rate
Θ: volume fractiion of the bulk soil containing water t: unit time Permeability (浸透性)Terms: permeable, adj 浸透(透過)される, permeate, vt -に浸透するDeficiency and evapotranspiration (etp)Ideal
|
Actual
Active transport (水能動輸送)
Materials: Barley roots |
[ stable isotope ( 安定同位体 )]
|
Def. transpiration (蒸散), Tp: the evaporation of water from plants
stomatal transpiration
Water-use efficiency, WUE (水利用効率)Def. WUE: the ratio of water used in plant metabolism to water lost by the plant through transpiration (at a unit time)a) instantaneous water-use efficiency (photosynthetic WUE)
ratio of the rate of carbon assimilation (photosynthesis, Ps) to the rate of transpiration
the ratio of biomass (dry weight, Dw) produced to the rate of transpiration |
Transpiration ratio (蒸散係数) = water requirement (要水量)
the amount of water required to produce a unit of plant biomass crop ≈ 200-1000: 200-1000 kg of water is requred to prodcue 1 kg of biomass Ex. To assimilate 1 g of organic substances, plants require water:Trees: 200-400 g / Herbs: 400-700 g |
|
1909 Soerensen: proposed the concept of pH A measure of the acidity or basicity of an aqueous solution, expressed by the concentration of hydrogen ion Def. ionic product of water (水のイオン積), Kw ≡ [H+]·[OH-]
→ depending only on temperature (T)
T (°C) → Kw = Potential hydrogen, or power of hydrogen pH is defined as minus the decimal logarithm of the hydrogen ion activity in a solution. So, pH is a dimensionless quantity. pH = -log10(H+) = log10(1/H+), where H+ is the activity of hydrogen ions.As wall as pH, we can define pOH = -log[OH-], and pKw = -logKw → pKw = pH + pOH = 14 |
Ex. Pure water: H2O ⇔ H+ + OH- → [H+] = [OH-] = 10-7
K = [H+][OH-]/[H2O] = 1.8 × 10-16 (mol) = 55.5 (mol) [H+][OH-] = 1.8 × 10-16 × 55.5 = 1.0 × 10-14, [H+] = [OH-] = 1 × 10-7 ∴ pH = log[H+] = 7
pH_____0____1____2_____3____4____5_____6____7 CH+ = r[H+]
r: activity coefficient (活量係数) Ex. 0.1 M HCl: -log[H+] = 1.09 = log-1(2.91) = 8.1 × 10-2, r = 0.81 |